 Graphene can double the electrical energy production of biofuel cells, as demonstrated by a group of researchers at the São Carlos Institute of Chemistry (IQSC) of the University of São Paulo (USP) and at the Federal University of the ABC (UFABC) in Santo André, in the São Paulo metropolitan region. Discovered in 2004 by Andre Geim and Konstantin Novoselov, at the University of Manchester, England, graphene, in addition to earning the researchers the Nobel Prize in Physics for their experiments, kindled a worldwide rush to use this new material. It is in the form of a sheet of carbon with atomic-scale thickness and interesting electrical, mechanical and optical properties.
Graphene can double the electrical energy production of biofuel cells, as demonstrated by a group of researchers at the São Carlos Institute of Chemistry (IQSC) of the University of São Paulo (USP) and at the Federal University of the ABC (UFABC) in Santo André, in the São Paulo metropolitan region. Discovered in 2004 by Andre Geim and Konstantin Novoselov, at the University of Manchester, England, graphene, in addition to earning the researchers the Nobel Prize in Physics for their experiments, kindled a worldwide rush to use this new material. It is in the form of a sheet of carbon with atomic-scale thickness and interesting electrical, mechanical and optical properties.
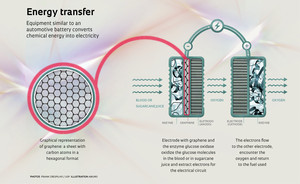
Brazilian researchers led by Professor Frank Crespilho, of IQSC-USP, demonstrated, in the cover article of the September 2014 issue of the journal Physical Chemistry Chemical Physics, that sheets of graphene oxide attached to flexible carbon fibers facilitate the transfer of electrons in biofuel cells. These devices convert chemical energy into electrical energy with the help of enzymes and can use the glucose in blood, for example, as fuel to provide the electricity needed by pacemakers or subcutaneous drug dispensers. Biofuel cells are an alternative energy source still restricted to laboratories. The biofuel cells developed in São Carlos are similar to batteries and have two flexible carbon fiber electrodes—the cathode (positive pole) and the anode (negative pole). They are one of the latest developments in energy source research. One of the fuel options for use with this device is sugarcane juice, which contains many sugars.

FRANK CRESPILHO / USPflexible carbon fiber used in electrodesFRANK CRESPILHO / USP
Biofuel cells can be microscopic or larger, and take the form of small plastic boxes. They may contain sugarcane juice and generate electricity and recharge mobile phone, tablet or laptop batteries. One cell can provide a voltage of a little over 1 volt (a AA battery, for example, furnishes 1.5 volt). Crespilho’s group has been working with these devices since 2010 (see Pesquisa FAPESP Issues nº 182 and 205). In an attempt to improve the electrical performance of the devices, the researchers placed graphene oxide sheets between the electrode and the enzyme glucose oxidase. This led to an at least two-fold increase in the transfer of electrons to the cell, resulting in doubled electricity production.
The electrons are released through the oxidation of glucose, which takes place on the surface of the anode, where the enzyme glucose oxidase, produced by the fungus Aspergillus niger, is placed. Thus, the electrons are transferred to the surface of the biofuel cell electrode, which uses them to generate electricity. This electron flow continues on to the other electrode, the cathode, where the oxygen is reduced. This process, known as redox, refers to oxidation (the loss of electrons) of the glucose and reduction (the gain of electrons) of the oxygen, both dissolved in the blood.
The presence of graphene creates a sort of bridge by reducing the distance between the center of the enzyme and the surface of the carbon electrodes, facilitating the passage of electrons. “We have already shown that it works better than carbon nanotubes because it makes better use of the enzyme’s properties. Recent studies have also shown that nanotubes can degrade the glucose oxidase, which does not happen when we use graphene,” says Crespilho, who is currently a visiting professor at the California Institution of Technology (Caltech). “I am part of a project attempting to understand how DNA and other biomolecules, like proteins, interact with the surface of other materials in terms of transferring electric charge,” he explains. “The idea is to strengthen this field of research in São Carlos and apply this knowledge to molecular bioelectronics in the future.”

FRANK CRESPILHO / USPCarbon fiber is exfoliated for production FRANK CRESPILHO / USP
German infrastructure
In addition to graphene oxide on flexible fibers, Crespilho is awaiting the construction of a biofuel cell with individual sheets of graphene that one of his IQSC students is developing at the Max Planck Institute in Germany. “It will surely be the thinnest biofuel cell ever built,” says Crespilho. “We don’t have the infrastructure in Brazil to build this device, but it should have two electrodes with a thickness of about a nanometer (one millionth of a millimeter),” he says. This is why PhD student Rodrigo Iost is working on building this new biofuel cell by the end of 2014, supported by a grant from FAPESP. “Last year, a thematic project [financed by FAPESP for four years] led by professor Osvaldo Novais, of the São Carlos institute of Physics (IFSC) was approved. This project will improve our infrastructure and allow us to produce new nanostructure films for biological applications. Then we’ll be able to build biodevices and employ them not only in biofuel cells, but also in implantable bioelectronic devices,” explains Crespilho. The molecular bioelectronics project developed by the group is also linked to the National Institute for Organic Electronics (Ineo-INCT), based at IFSC.
Projects
1. Interaction between biomolecules and cellular systems with 0D, 1D and 2D nanostructures using electrochemical methods (nº 2009/15558-1); Grant Mechanism Regular research project; Principal Investigator Frank Crespilho (USP); Investment R$92,262.80 and $50,821.57 (FAPESP).
2. Bioelectrochemical study of oxidoreductase enzymes immobilized in 1D and 2D nanomaterials (nº 2013/15433-0); Grant Mechanism Scholarship abroad — Regular — Research Internship — PhD; Principal investigator Frank Crespilho (USP); Grant Recipient Rodrigo Iost (USP); Investment R$93,415.01 (FAPESP).
3. Nanostructured films from biologically relevant materials (nº 2013/14262-7); Grant Mechanism Thematic Project; Principal investigator Osvaldo Novais (USP); Investment R$1,150,950.14 (FAPESP).
Scientific article
MARTINS, M.V. A. et al. Evidence of short-range electron transfer of a redox enzyme on graphene oxide electrodes. Physical Chemistry Chemical Physics. v. 16 n. 33 p. 17349–18044. set. 2014.