
léo ramos chaves
Perovskite solar cells developed in the Nanotechnology and Solar Energy Laboratory in the Institute of Chemistry at the University of São Pauloléo ramos chavesA new generation of solar cells created from synthetic crystalline material are known as perovskite solar cells. They were chosen as one of the top ten emerging technologies at the 2016 World Economic Forum, a Swiss event that brings together business leaders and politicians every year to discuss global issues. The material has garnered excitement among scientists due to its high capacity for converting photons into electrons, thus generating electricity. In July of this year, the Ulsan National Institute of Science and Technology (UNIST) in South Korea announced its laboratory-scale production of perovskite solar cells with a record energy efficiency of 22.1%. This rate, obtained for small cells (those smaller than the cells used in commercial models), surpassed that of the silicon panels that dominate the market. (Silicon solar panels currently represent 90% of sales.) The silicon cell conversion rate currently holds between 15% and 20%, depending on the purity of the silicon used to build the solar panels.
Solar panel manufacturers and numerous research groups around the world, including in Brazil, are working to improve this technology. They still need to overcome certain obstacles, such as its low durability, before it can reach the consumer market. Oxford Photovoltaics, part of Oxford University in the United Kingdom, has built perovskite solar cells that exhibit 20% efficiency. The researchers are now working to apply them to silicon panels to increase the panels’ rate of energy conversion. The founder of the company, a physicist named Henry Snaith, was one of the first scientists to recognize the potential of the material as having the ability to convert light from the sun into electricity. Oxford Photovoltaics hopes to launch the first commercial models of these cells by the end of 2018, company president Frank Averdung told Bloomberg News Agency in March.
“Perovskite solar cells are a recent and promising technology,” says chemist Rodrigo Lopes Sauaia, chief executive of the Brazilian Photovoltaic Solar Energy Association (ABSOLAR), an institution that brings together companies from the sector. “These are great innovations and developments in the photovoltaics sector, especially in Brazil, since the country has such enormous potential for generating solar energy,” he says (see Pesquisa FAPESP, issue No. 258). Brazilian production of this type of energy is still limited to 176 MW, which corresponds to 0.1% of the country’s energy matrix. However, this percentage has been increasing rapidly. “It’s likely to reach 1 gigawatt by the end of the year,” reports Sauaia. In June, the Lapa Solar Park began operations in the semi-arid sertão region of Bahia State. It is the largest power station in Brazil, with the capacity to produce 158 MW—enough to meet the needs of 166,000 households per year.
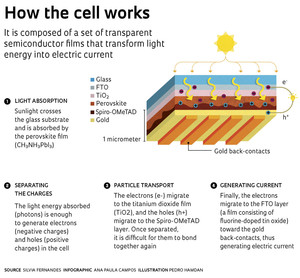
The perovskite used in solar cells functions as a semiconductor with the chemical formula CH3NH3PbI3. Its structure is similar to that of calcium titanate (CaTiO3), which was first discovered in the Ural Mountains in Russia. The mineral was named after the Russian mineralogist Lev Aleksevich von Perovski (1792–1856). The raw material used to produce the cells is not taken from nature, but is produced synthetically in the lab. The cells are built in layers, with different thin films that each have a unique function and chemical makeup (see infographic).
 The rapid evolution of this technology has caught scientists’ attention. When perovskite was applied to solar cells for the first time in 2009, its rate for converting light into electric energy was less than 4%. Less than a decade later, this rate grew more than fivefold (see graph below), and the trend is likely to continue. Professors Yang Yang of the University of California, Los Angeles, and Jingbi You from the Semiconductors Institute at the Chinese Academy of Sciences estimate that these solar cells are likely to reach 25% efficiency within two years, according to the article published in Nature in April. Meanwhile, silicon wafers have been on the market for more than 50 years and seem to have reached their limit. No significant progress has been reported in their rate of conversion in the last 15 years.
The rapid evolution of this technology has caught scientists’ attention. When perovskite was applied to solar cells for the first time in 2009, its rate for converting light into electric energy was less than 4%. Less than a decade later, this rate grew more than fivefold (see graph below), and the trend is likely to continue. Professors Yang Yang of the University of California, Los Angeles, and Jingbi You from the Semiconductors Institute at the Chinese Academy of Sciences estimate that these solar cells are likely to reach 25% efficiency within two years, according to the article published in Nature in April. Meanwhile, silicon wafers have been on the market for more than 50 years and seem to have reached their limit. No significant progress has been reported in their rate of conversion in the last 15 years.
Degree of purity
Perovskite cells are also cheaper and easier to produce than silicon cells. “To obtain high energy efficiency, silicon cells need to have a very high degree of purity, which increases energy consumption during manufacturing and raises their cost,” explains chemist Ana Flávia Nogueira, a professor at the Institute of Chemistry of the University of Campinas (UNICAMP) and leader of one of the research groups studying this type of device in the country, the Nanotechnology and Solar Energy Laboratory (LNES). Silicon purity is necessary because small imperfections in the crystal interfere with its ability to transform absorbed light into electricity.
Perovskite solar cells, however, do not require a high degree of purity, since defects in their structure do not reduce their efficiency. They are made with cheap chemical compounds and can be created using simple, cost-effective methods. What’s more, the production process does not contribute to global warming. During the production of the silicon wafers, silicon dioxide (SiO2), the basic raw material used in the device, must be melted at high temperatures (approximately 1,500°C). This process releases carbon dioxide (CO2) into the atmosphere. “The manufacturing of perovskite cells does not emit CO2,” says Nogueira.
Nogueira’s group was the first to produce perovskite solar cells in Brazil in 2016. “This study began with the master’s thesis by chemist Rodrigo Szostak. It was not difficult to start development of these cells, since our laboratory has been researching organic solar cells and dye-sensitized solar cells since 2004, two technologies which inspired perovskite cells,” says the researcher. The product developed in the LNES has already reached rates of efficiency close to 16% and is likely to reach 18% by the end of the year.
Another feature of perovskite solar cells is their thickness: approximately 1 micrometer (one millionth of a meter), which is much smaller than the 180-micrometer thickness of silicon wafers. “They are produced in the form of ultra-thin films and can be semitransparent, which can aid in the manufacturing of light, flexible panels, thus allowing for a greater number of applications,” says chemist Silvia Letícia Fernandes, who earned a PhD on the subject. One of the problems of silicon photovoltaic cells is that they are heavy and rigid, which hinders and limits the types of places where solar panels can be installed.
 At São Paulo State University (UNESP) last year, Fernandes defended her doctoral dissertation titled “Development of Perovskite Solar Cells Based on Nanostructured Oxide Films.” She was advised by Dr. Maria Aparecida Zaghete from the Institute of Chemistry of UNESP’s Araraquara campus. The preparation of solar cells in the study was supported by Dr. Carlos Frederico de Oliveira Graeff from the School of Sciences of UNESP’s Bauru campus. Zaghete and Graeff are researchers from the Center for the Development of Functional Materials (CDMF), one of the Research, Innovation, and Dissemination Centers (RIDC) supported by FAPESP.
At São Paulo State University (UNESP) last year, Fernandes defended her doctoral dissertation titled “Development of Perovskite Solar Cells Based on Nanostructured Oxide Films.” She was advised by Dr. Maria Aparecida Zaghete from the Institute of Chemistry of UNESP’s Araraquara campus. The preparation of solar cells in the study was supported by Dr. Carlos Frederico de Oliveira Graeff from the School of Sciences of UNESP’s Bauru campus. Zaghete and Graeff are researchers from the Center for the Development of Functional Materials (CDMF), one of the Research, Innovation, and Dissemination Centers (RIDC) supported by FAPESP.
The achille’s heel
Even with so many advantages, perovskite cells still need to overcome certain challenges before they can become a commercial product that can compete for space in the solar power market. The main issue is the material’s limited durability. “It’s the Achilles’ heel,” admits chemist Rodrigo Szostak, from the UNICAMP research group. “Another significant problem is its limited stability relative to that of silicon. Perovskite is sensitive to water and moisture, both of which degrade it,” he explains. The first cells ever made with this material remained stable for only a few minutes, but changes to the material’s structure have increased its durability to just over one year.
Recently, Dr. Michael Gratzel of the Federal Polytechnic School of Lausanne (EPFL) in Switzerland, one of the experts on the study of perovskite cells, manufactured 10×10 cm2 solar panels using a process that had been adapted for industrial production. The devices exhibited 11.2% efficiency and remained stable for more than 10,000 hours (415 days). Despite the progress, this amount of durability is still much lower than that of silicon panels, which can be used without degradation for up to 25 years.
Another problem to overcome is the use of lead in cell assembly. Lead is a chemical element that creates risk for both human health and the environment. “Lead is always a significant environmental concern, but the quantity used here is very small. Its use in solar cells in the formation of very thin films would have much less of an impact on the environment than the lead-acid batteries used by the automobile industry,” notes Fernandes. Problems could be avoided through the adequate disposal of panels and the use of the cells in secure contexts. “The use of solar panels in power stations and on previously prepared land puts them at a low risk of causing environmental damage,” Nogueira explains.

ENEL / MARKETING MATERIALS
The Lapa Solar Park in Bahia, the largest solar power station in Brazil, generates enough energy to meet the needs of 166,000 households per yearENEL / MARKETING MATERIALSMore stable cells
To help circumvent the low stability of perovskite cells, the UNESP group inserted films made of niobium pentoxide (Nb2O5) into the cell composition, which made it more stable. “Solar cells basically consist of a perovskite film and two other films, one which is responsible for electron transport and the other of which is responsible for holes [a particle characterized by the absence of an electron and which has a charge of the same value but a positive sign rather than a negative sign]. These electrons and holes generated by light migrate to opposite sides of the material, creating tension that can be used to power electrical devices,” explains Fernandes. “Most cells use titanium dioxide [TiO2] for electron transport. We introduced niobium pentoxide, which proved to be very efficient and which even improved stability.”
The device created at UNESP exhibited rates of efficiency as high as 15%. Part of the study was performed at the Swiss Federal Laboratories for Materials Science and Technology (EMPA) under the guidance of Dr. Frank Nüesch. “In 2015, I spent five months in Dr. Nüesch’s lab. He provided us with the physical space and experience necessary for assembling the cells. We used the niobium pentoxide films prepared in Brazil and assembled the cells in Switzerland. Today, we are able to assemble and characterize the devices in the Laboratory of New Materials and Devices (LNMD) [in Brazil] at the same level of quality,” says Fernandes.
A third Brazilian research group from the Federal University of Santa Catarina (UFSC) is also working to improve perovskite solar cells. Their innovation was to add a titanium dioxide aerogel to the architecture of the device in order to raise its energy conversion rate. The research was led by physicist Carlos Rambo and his team. Rambo is coordinator of the Laboratory of Electrical Materials (LAMATE) of the Department of Electrical and Electronic Engineering. The work included partnership with physicists Maria Luísa Sartorelli and Françoise Toledo Reis from the Laboratory of Nanostructured Systems (LABSIN) of the Department of Physics.

Sílvia Fernandes / Marketing Materials
Material created by the UNESP Araraquara group has achieved 15% energy efficiencySílvia Fernandes / Marketing Materials“Aerogel is a material known as solid smoke because it has a large surface area and is very light. We were the first in the world to develop perovskite solar cells based on aerogel,” says Rambo. “We added the titanium dioxide aerogel to the device’s architecture and doubled its efficiency relative to that of a compact cell.”
For Sauaia from ABSOLAR, research efforts in Brazil and around the world are fundamental for improving the physical and chemical characteristics of perovskite cells and for making headway in the cell production process. “The current challenge is to turn small cells, which have produced good results in the lab thus far, into commercial products produced on a large scale,” says Sauaia. According to Sauaia, the success of this new technology will depend on the existence of a competitive solar panel that meets the requirements of the market.
Greater absorption power
A team from MIT uses carbon nanotubes and nanophotonic crystals to create a more efficient solar device
A group of researchers at the Massachusetts Institute of Technology (MIT), led by mechanical engineer Evelyn Wang, physicist Marin Soljacic, and doctoral candidate David Bierman, are working on a new type of solar cell that they say is able to generate twice as much power as existing silicon panels. The secret of the new technology is its high capacity for absorbing solar radiation, according to MIT Technology Review. Modern photovoltaic cells absorb only wavelengths of light in the visible range, from violet to red; the rest is lost. The device is able to absorb energy from across the solar spectrum for the generation of electricity.
Another advantage is that this type of cell could also be efficient on days with low sunlight. Although it depends on solar radiation to generate electricity, once the absorbing material has captured that light, it can generate heat. This heat can be stored to produce energy on cloudy days or even at night. In the laboratory, the prototype exhibited a relatively low efficiency index of 6.8%, but its inventors believe that it has the potential to evolve.
Some obstacles still need to be overcome, such as the high cost of manufacturing the new system. Another challenge is proving that the technology is viable under normal environmental conditions, since all tests thus far have been performed in a vacuum and not in the environment. The new device was described by MIT Technology Review as a promising technology.
Projects
1. Hybrid nanostructures in third-generation (3G) solar cells (No. 14/21928-4); Grant Mechanism Regular Research Grant; Principal Investigator Ana Flávia Nogueira (UNICAMP); Investment R$291,986.83.
2. Development of hybrid solar cells based on nanostructured ZnO and Nb2O5 films (No. 12/07745-9); Grant Mechanism PhD Grant – Brazil; Principal Investigator Maria Aparecida Zaghete Bertochi (UNESP); Scholarship Beneficiary Silvia Letícia Fernandes (UNESP); Investment R$106,393.98 and R$45,284.17 (Internship Grant for Research Abroad, No. 14/23336-7).
Scientific articles
SZOSTAK, R. et al. Understanding perovskite formation through the intramolecular exchange method in ambient conditions. Journal of Photonics for Energy. V. 7, No. 2. 24 May 2017.
FERNANDES, SL et al. Nb2O5 hole blocking layer for hysteresis-free perovskite solar cells. Materials Letters. V. 181. 15 Oct. 2016.
PINHEIRO, G. K. et al. Increasing incident photon to current efficiency of perovskite solar cells through TiO2 aerogel-based nanostructured layers. Colloids and Surfaces A: Physicochemical and engineering aspects. V. 527, p. 89-94. 20 Aug. 2017.