A versatile metal, abundant in Brazil, niobium is gradually finding applications beyond common and specialized metal alloys. The material is now making strides in new technological territories, with applications ranging from electric vehicle batteries to acne creams, as well as fungicides and catalysts. In November last year, the Brazilian Metallurgy and Mining Company (CBMM), the global leader in the manufacture and sale of industrialized metal products, opened a factory in Araxá, in the state of Minas Gerais, in partnership with British company Echion Technologies, to produce anodes—components of lithium-ion batteries—from niobium. Also produced in Minas Gerais, a new product containing niobium was launched in July 2024 and has made its way onto pharmacy shelves. It is an anti-acne cream with an ingredient created using nanotechnology patented by Nanonib, a spin-off from the Federal University of Minas Gerais (UFMG).
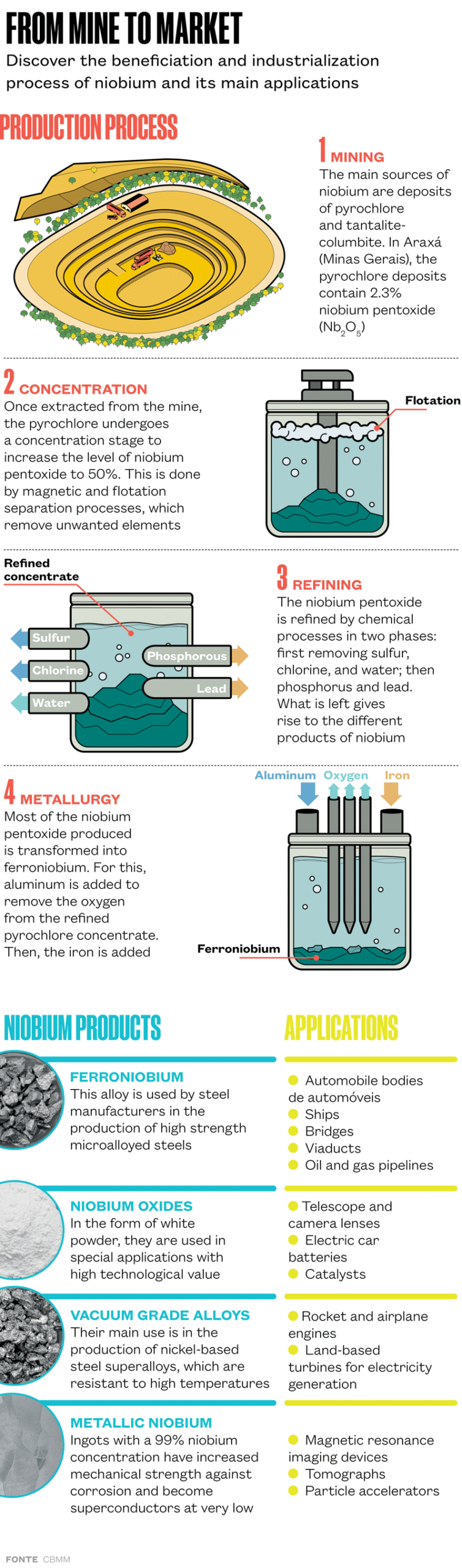
The new applications differ from the long-established uses of the material. Brazil stands out as the largest producer and owner of niobium reserves, with around 90% of global production. The largest extractable deposit of pyrochlore—a mineral composed of up to 71% niobium oxide—is in Araxá and is operated by CBMM, which is owned by the Moreira Salles family (see Pesquisa FAPESP issue nº 277).
Since the 1970s, niobium has been used for manufacturing special steels used in oil and gas pipelines, since it provides more security to the welded regions, and in the automotive industry. It is used as a raw material by companies in the construction sector for structures of bridges and large buildings.
“Between 80% and 90% of what we sell are ferroniobium based products, a metallic alloy composed of niobium and iron,” says materials engineer Rafael Mesquita, director of technology at CBMM. “The main advantage is that niobium increases the mechanical strength of steels, providing a stronger product and increasing security. As a result, it is possible to reduce the amount of steel in the final product, thus reducing CO₂ [carbon dioxide] emissions.”

Léo Ramos Chaves / Revista Pesquisa FAPESPNiobium and its different appearances: in the middle, the metallic alloy of ferroniobium, which increases the mechanical strength of steelsLéo Ramos Chaves / Revista Pesquisa FAPESP
Another already well-established use is in the production of superalloys—not based on iron, but on other metals, especially nickel. This material is resistant to extremely high temperatures and is heavily consumed by the aerospace industry in the manufacture of airplane turbines. It is also used in the production of turbines for power generation.
Superconducting alloys manufactured with niobium are present in the magnets of the Large Hadron Collider (LHC), the European Organization for Nuclear Research’s (CERN) particle accelerator, and in magnetic resonance imaging devices. “The wire that produces the magnetic field for these pieces of equipment is made from a niobium-titanium alloy,” says Mesquita, who had funding from FAPESP on his master’s degree focused on aluminum alloys at the Federal University of São Carlos (UFSCar). “The applications of niobium on its own are limited and not very significant. But the situation changes when it is combined and added to other materials.”
Real-life vibranium
One of the Brazilian research groups in the field is coordinated by chemist Luiz Carlos Alves de Oliveira, of the Department of Chemistry at UFMG, who has been studying the properties and applications of niobium since 2006. He estimates that the knowledge has so far generated up to a dozen patents and around 70 scientific articles. “We usually say that niobium is the real-life vibranium,” says Oliveira, lightheartedly, referring to the fictional material with special powers portrayed in Marvel movies, such as Black Panther.
The chemical structure of niobium, explains the chemist, is responsible for the versatility and the multiplicity of applications. “Its most important unit resembles a tetrahedron [a geometric shape with four triangular faces], and the manner in which these smaller units join and how it accommodates the neighboring atoms is what makes it so special,” highlights Oliveira.
Specialized in the surface chemistry of materials, the researcher and his team noticed that it was possible to modulate the oxidizing property of the surface of niobium oxide, a chemical compound derived from niobium, containing oxygen, used for a range of applications (see infographic). “Depending on how we obtained its chemical form, the oxygen became highly reactive, capable of detaching from the surface. And it is this oxygen, for example, that is capable of killing bacteria.”
Oliveira says that he did tests with other similar chemical elements, but only niobium displayed this property. “We have done countless tests over the years. There are chemical elements that retain this special reactive oxygen, but the material loses activity very quickly. They work in basic research, but not for becoming a product.”
In the wake of the COVID-19 pandemic, the group created a liquid spray based on niobium to combat the Sars-CoV-2 virus, called Innib-41, to be sprayed on the hands. “The liquid forms a film or a nanofilm as if it were an invisible glove. When it encounters a virus or a bacterium, the oxygen detaches from the niobium and reacts with the cellular wall, causing a type of suffocation of the microorganism,” he explains.
The discovery that the niobium molecule killed bacteria arose when the researchers were developing a tooth whitener. According to an article published by the team from UFMG in Clinical Oral Investigations in 2023, the modification of the niobium oxide structure through its association with hydrogen peroxide not only whitened the teeth but also killed the bacteria responsible for cavities. The group ended up creating a startup, called Nanonib, to obtain patents and license products.
Although the spray was approved by ANVISA, the group has chosen not to manufacture it and to focus on other products first instead. The first to go on sale that uses this same technology—with an active ingredient called Blue Active—was launched in July 2024. It is the anti-acne cream Acnano. “We made a partnership with the cosmetics manufacturer from Minas Gerais, Yeva, responsible for manufacturing and selling the products of this line.”
The startup is currently conducting field tests for a third line of products aimed at agribusiness. “We have submitted patent applications to the relevant authorities for two molecules. A third is also being studied,” states Oliveira. They are two fungicides, one for use on seeds and the other for leaves, already in testing. The three products for the agricultural sector use the same principle as the Innib-41 virus spray: a film of niobium is formed on the surface of the leaves or seeds.
Photocatalysts
At the Aeronautics Institute of Technology (ITA), chemist André Esteves Nogueira, from the Department of Chemistry, conducts research related to niobium applications in two areas. One of them is linked to photocatalysts, focused on the conversion of CO₂ into products with more added value, such as ethanol, methanol, acetic acid, and methane—photocatalysts are materials that, once activated by light, accelerate chemical reactions.
The second path being researched, with funding from FAPESP, is aimed at the production of hydrogen for power generation. “We are testing niobium as a catalyst in the process of sodium borohydride hydrolysis,” says Nogueira. According to the chemist, sodium borohydride may offer advantages in the production of hydrogen because it is a solid—a white salt, sold in powdered form—which would facilitate transportation and allow for on-site production.
“The idea is to buy the borohydride, which has a great hydrogen storage capacity, take it to the location where energy will be generated, dissolve it, and add the catalyst. All of this needs an external energy source, as occurs in the traditional process of manufacturing green hydrogen, via water hydrolysis,” says the researcher from ITA.

YEVA LaboratoiresA line of cosmetics with niobium-based nanotechnology created in the UFMG laboratoriesYEVA Laboratoires
To accelerate this reaction, so that it can be used on an industrial scale, the researcher and his students are studying variations of niobium catalysts modified with cobalt. “An article published in the International Journal of Hydrogen Energy describes the modifications that we have made with nanoparticles of cobalt on the surface of the niobium oxide.” The results, Nogueira says, were promising. “Now we are trying to perfect the catalyzing activity using different sources of cobalt.”
Niobium also has an important role in the manufacture of components for orthopedic implants. When added to titanium, this element, besides being biocompatible, makes it possible to create materials with increased mechanical strength and elastic properties that minimize harmful effects caused by certain types of implants in the human body.
Mechanical engineer Rubens Caram, from the University of Campinas (UNICAMP), is coordinating a FAPESP-funded research project aimed at applying additive manufacturing techniques—also known as 3D printing—in the preparation of titanium- and niobium-based components with optimized properties.
In recent years, Caram’s team have published dozens of scientific articles related to the effect of niobium on titanium alloys, always with support from CBMM. One of them, produced in partnership with the University of North Texas, in the USA, focuses on the design of fixation plates for bone repair with optimized property gradients and mechanical behavior using titanium alloys containing niobium.
In Araxá, CBMM has had a program aimed at promoting new uses of niobium for years. The company has invested R$250 million annually in technological research and development (R&D), with 90% of this amount in an open innovation model with other companies, universities, and research centers. “We have a much greater production capacity than the total demand of the niobium market. The limiting factor in our business are the applications,” says Mesquita.
A large portion of the amount invested in R&D in the past months is aimed at the study of batteries. The new unit for niobium-based anode production—a technology patented by Echion called XNO—has the capacity to produce 2,000 tons of material per year, equivalent to 1 gigawatt of lithium-ion cells. The XNO technology, according to the company from Minas Gerais, increases the efficiency and durability of batteries, enabling ultrafast and safe charging.
At the CBMM facilities, a demonstration electric bus has been circulating since last June powered by lithium-ion batteries with niobium developed in partnership with Toshiba. Instead of the conventional eight hours or more to recharge, the process takes just eight minutes.
The company is taking part in a project coordinated by the Institute of Technological Research (IPT) in São Paulo, aimed at developing new alloys of materials, including niobium, to produce metallic components using additive manufacturing. The initiative, funded by FAPESP, has participation from other institutions in the state.
“The idea is to start with a metallic powder containing niobium and arrive at a final part through additive manufacturing, instead of using the conventional manufacturing process,” explains the director of CBMM. “Take the example of an automotive component. To manufacture it, it is necessary to produce the steel, make a steel bar, shape the steel part, drill it, etc. It’s a long process. With additive manufacturing, we can start with the powder and quickly arrive at the final part.”
The story above was published with the title “New frontiers for niobium” in issue in issue 348 of february/2025.
Projects
1. Production chain development for metal components by additive manufacturing (nº 20/06984-6); Grant Mechanism Problem-Oriented Research Centers in São Paulo; Principal Investigator Mário Boccalini Júnior (IPT); Investment R$3,846,662.90.
2. Rapidly solidified AI alloys: Microstructures and properties (nº 00/00218-6); Grant Mechanism Master’s Fellowship; Supervisor Walter José Botta Filho (UFSCar); Beneficiary Rafael Agnelli Mesquita; Investment R$2,910.00.
3. Development of multifunctional niobium catalysts: Advances in CO2 conversion and H2 generation (nº 23/17686-4) Grant Mechanism Regular Research Grant; Principal Investigator André Esteves Nogueira (ITA); Investment R$267,313.49.
4. Titanium alloys: The use of phase transformations and additive manufacturing to obtain materials with functional gradients (nº 18/18293-8); Grant Mechanism Thematic Project; Principal Investigator Rubens Caram Junior; Investment R$5,576,609.50.
Scientific articles
GRIFFITH, K. J. et al. Titanium niobium oxide: From discovery to application in fast-charging lithium-ion batteries. Chemistry of Materials. Vol. 33, no. 1. Dec. 2020.
CANESCHI, C. S. et al. Bleaching effectiveness and cytotoxicity of new experimental formulation of niobium‑based bleaching gel. Clinical Oral Investigations. Vol. 27, no. 4. pp. 1613–21. Apr. 2023.
LIMA D. D. et al. Laser additive processing of a functionally graded internal fracture fixation plate. Materials & Design. Vol. 130, pp. 8–15. Sept. 15, 2017.
VALENTIN, M. et al. Investigating multi-material Ti-42Nb lattice structures fabricated via laser powder bed fusion using a genetic algorithm to optimize Ti-5553 reinforcement band position. Materials Science and Engineering: A. Vol. 925, 147833. Mar. 2025.
SANTOS, F. L. et al. Hydrogen generation via NaBH4 hydrolysis over cobalt-modified niobium oxide catalysts. International Journal of Hydrogen Energy. Vol. 92, pp.113–23. Oct. 2024.
BRUZIQUESI, C. G. O. et al. Nióbio: Um elemento químico estratégico para o Brasil. Química Nova. Vol. 42, no. 10. Oct. 2019.
Republish