
ZAK HUSSEIN / CORBIS / GETTY IMAGES
Trois hommes qui marchent, Alberto Giacometti, bronze with brown patina, 1948ZAK HUSSEIN / CORBIS / GETTY IMAGESFive years ago, surgeon Paulo Alcântara was intrigued to see that two patients of the same age and with the same type of advanced bowel cancer who had been treated during the same week at the University Hospital of the University of São Paulo (HU-USP) responded differently to the same treatment. One was obese, while the other was very thin. The thin patient’s lack of body mass was a manifestation of cachexia, a syndrome characterized by continuous loss of muscle mass and appetite that can accompany (and worsen) not only cancer, but also AIDS, heart failure, and chronic obstructive pulmonary disease (COPD). Seen in 40% of people with cancer and 80% of patients hospitalized with malignant tumors, cachexia makes treatment difficult and is associated with 20% of the deaths caused by this disease. The thin patient died a year and a half after treatment as a result of cancer and cachexia, while the other patient lived for another four years.
Intrigued by this situation, Alcântara sought the help of biologist Marília Seelander, who has been studying physical exercise, inflammation, and cancer at the USP Institute of Biomedical Sciences (ICB-USP) for 25 years (see Pesquisa FAPESP, issue No. 89). Based on the results of experiments in animal models, the two researchers planned a study to evaluate the possible effects of physical activity on people with cancer and cachexia.
Preliminary results from the tests performed within the University Hospital indicate that a physical exercise regimen—in this case, walking or running on a treadmill for one hour a day for six weeks—can reduce the inflammatory processes that result in weight loss in cases of cachexia. Participants with cancer and cachexia who exercised recovered muscle mass and appetite and exhibited better postoperative recovery than those without cachexia. There was also a change in the patients’ cytokine profiles. Cytokines are proteins that activate defense cells. In these patients, levels of proinflammatory cytokines, which cause and worsen cachexia, were found to have fallen, while anti-inflammatory cytokine levels rose.
So far, 332 patients with cancer of the stomach, pancreas, or intestine—some of whom experienced cachexia—participated in the study; 272 were placed in the sedentary group, and 50 underwent physical training. “The blockage of cachexia could allow for a more intensive treatment, improve quality of life, and increase patient survival,” says Alcântara. “However, we have to reach 100 cases in each group of patients—with and without cancer and with and without cachexia—to produce statistically significant results.” The ongoing studies, proposed in a 2015 article in the journal Current Opinion in Supportive and Palliative Care, integrate teams from USP, the São Paulo State Cancer Institute (ICESP), Santa Casa Hospital of São Paulo, and Santa Marcelina Hospital.

LÉO RAMOS CHAVES
USP’s University Hospital exercise room used to help patients regain weightLÉO RAMOS CHAVESScientific research has yet to be completed on the effect of physical exercise on people with cancer and cachexia, but its effects have been observed in people with COPD. A group made up of researchers from Germany and the Netherlands found that four months of intense exercise contributed to improved health and muscle strength in people with COPD and cachexia relative to patients who received nutritional supplements and to controls. The study included 81 patients and was published in June of this year in the Journal of Cachexia, Sarcopenia and Muscle.
“The metabolic changes characteristic of cachexia weaken the body, favor tumor growth, and make treatment difficult,” Alcantara explains. On June 13, he attended a meeting at ICB-USP, where researchers from Seelander’s team presented the results of the blood and tissue analyses performed on the subjects from the University Hospital. Individuals with cachexia exhibited a reduction in protein metabolism and in the levels of two hormones: leptin, which regulates appetite, and insulin, which aids in glucose uptake by cells. The functioning of the hypothalamus, a region of the central nervous system that controls hunger, was found to have been affected. Bacterial communities in the gut, which may cause or aggravate inflammatory processes, were found to have changed, and the muscles, including that of the heart, were found to have weakened. “The imbalance caused by cachexia is so intense that people continue to lose weight, even when they receive nutritional supplementation, because the cells can no longer absorb the nutrients,” says Seelander.
Cachexia was first recorded by the Greek physician and philosopher Hippocrates (460–370 BC) and was described as a “profound degradation” by the French physician Paul Broca (1824–1880) in his treatise on tumors published in 1866. Brazilian physician Alfredo Leal Pimenta Bueno (1886–?) presented cachexia as one of the signs of the final stage of cancer, and he analyzed its origins in a series of articles published in the journal Brazil Médico from 1926 to 1928.
“Cachexia favors tumor growth and may progress to the point of becoming irreversible,” notes oncologist Gilberto de Castro Jr. of ICESP. Treating the tumor does not always cure cachexia. “Physical exercise is somehow able to block it and serve as supportive therapy,” he says. “We still have to determine the most adequate intensity, frequency, and duration of exercise, but we need to get cancer patients to move more.” Several studies have already indicated the benefits of physical exercise in the fight against cancer.
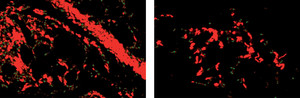
MIGUEL LUIS BATISTA JR. / UMC
Collagen protein production (in red) intensifies in people with cancer and cachexia; it results in the formation of fibers that impair the functioning of fat cells (left). Patients with cancer and without cachexia have less collagen (right)MIGUEL LUIS BATISTA JR. / UMCIn the laboratory
“Physical exercise minimizes cachexia,” agrees physical education professor Patrícia Chakur Brum, whose conclusion is based on her experiments with animal models at the School of Physical Education and Sports at USP. She and her team used two groups of mice with Walker 256 tumors; these tumors are used in experimental studies because they grow rapidly and induce the muscular atrophy characteristic of cachexia. One group included sedentary animals, and the animals in the other group exercised on a treadmill daily for 15 days starting the day after the tumor cells were injected. The animals that exercised had 31% higher survival rates than those in the sedentary group, though there was no reduction in the rate of progression of the tumor, which is an aggressive type.
Brum’s findings suggest that aerobic exercise can improve the functioning of muscle cells in animals with Walker 256 tumors. “Physical exercise may not directly affect the tumor, but it does render the muscles more functional,” she says. Her group has also found that previous physical activity may delay the onset of skin and breast tumors in mice. In Seelander’s laboratory at ICB-USP, the results were more dramatic: in the animals that exercised on a treadmill or swam as part of a longer exercise program, the size of the Walker 256 tumor decreased by 50%.
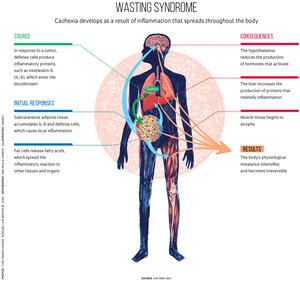
Though the loss of muscle is the most visible manifestation of cachexia, it is not the cause; it is actually one of the consequences of the process in which the body consumes itself. “We still do not know how or when cachexia begins,” acknowledges physical education expert Miguel Luiz Batista Júnior, a professor at the University of Mogi das Cruzes (UMC). The onset of the syndrome is likely to be inflammation triggered by an intense production of both proinflammatory cytokines and interleukin-6 (IL-6), a process which occurs as a result of the actions of defense cells against tumors. “The level of IL-6 circulating in the bloodstream increases two- to threefold in people with cancer, and levels are five to six times higher [than normal] in patients with cachexia,” says Batista, who has studied the mechanisms of cachexia since 2008 as part of a collaboration between USP and the University of Boston in the United States (see infographic).
 Other strategies
Other strategies
Based on tissue samples from cancer patients, the teams from UMC and USP concluded that subcutaneous white adipose tissue atrophies and becomes fibrous as a consequence of the accumulation of defense cells and the formation of an external mesh of collagen proteins on adipocytes, as detailed in a 2016 article in the Journal of Cachexia, Sarcopenia and Muscle. “As a result, adipose tissue loses its ability to store energy for the body,” says Batista.
In his laboratory, Batista tested pioglitazone, a medication which has been used to treat diabetes, to see whether it could slow or stop the progression of cachexia. The drug hindered muscle mass reduction and increased survival of Walker 256 tumor rats by 27% relative to the controls. Published in PLOS One in 2015, the study argues that pioglitazone could be effective in both early and late stages of cachexia by reducing insulin resistance and facilitating glucose uptake by cells, though it may cause damage to the heart. Current clinical trials in the United States are also assessing the possible uses of metformin, another antidiabetic drug, as well as other medications such as ghrelin and anamorelin.
At the Institute of Biology of the University of Campinas (IB-UNICAMP), biologist Maria Cristina Marcondes found that leucine, an amino acid with anti-inflammatory properties, prevents muscle degradation in Walker 256 tumor rats. “The tumor continues to grow, but animals regain at least 25% of muscle mass,” she explains. Her group is also looking for molecular markers that can be used to detect the onset of this syndrome. This information is useful for medical teams that treat people with cancer and cachexia and that would like to make a diagnosis and act as soon as possible to avoid weight loss and physiological imbalance.
Seelander notes that, because the measurement of IL-6 and other inflammatory cytokines is expensive, an alternative would be to examine both C-reactive protein (CRP) levels in the liver and hemoglobin and albumin levels in the blood. According to Seelander, high CRP levels or albumin and hemoglobin levels well below normal could indicate the onset of cachexia before loss of muscle mass occurs. In addition, clear spots seen on the muscles in CT scans could indicate the infiltration of fat or adipose tissue cells, signaling the onset of an inflammatory process that can lead to muscle loss.
As the research evolves, these proposals are likely to help stop a problem that is typically not detected until the loss of muscle mass is already evident. In the coming years, cachexia treatment may combine several strategies, such as the use of dietary supplements, physical exercise, and new drugs, in an attempt to prevent the physiological imbalances that contribute to the progression of cancer and other diseases. Meanwhile, Paulo Alcântara is still driven by the unanswered question that has guided his research for the last five years. So far, there are only guesses as to why some people experience cachexia and others do not, even when they are the same age and in the same stage of cancer.

LÉO RAMOS CHAVES
Aerobic exercises like running and cycling are recommended to help prevent cancer and to reduce the unwanted effects of medicationsLÉO RAMOS CHAVESWorking out: Part of prevention
Physical exercise helps prevent cancer, aids in recovery after surgery, and reduces the side effects of medications, the recurrence of tumors, and mortality rates, according to studies performed in the United States. In a June 2016 article in JAMA: The Journal of the American Medical Association, Steven Moore and his epidemiology team at the National Cancer Institute in the United States presented a meta-analysis of 12 studies from the United States and Europe that considered the effects of physical activity on 26 types of cancers in 1.4 million people over the course of 11 years. Researchers associated moderate or intense physical activity (such as walking) during leisure time with a lower risk of 13 types of cancer, as well as with a 42% drop in esophageal tumors and a 10% decrease in breast tumors, even among obese people and smokers.
If doctors and people with cancer were to adopt physical exercise as part of treatment, they would be following the same pattern witnessed years ago in the treatment of heart disease, according to Carlos Eduardo Negrão, a professor at the School of Physical Education and Sports of the University of São Paulo School of Medicine and director of the Cardiovascular Rehabilitation and Exercise Physiology Center of the Heart Institute (InCor), also within the University of São Paulo (USP). “Until the 1970s, people with heart failure were advised not to exercise; later, it became a more common recommendation, and it is now an important part of treatment,” he explains (see Pesquisa FAPESP, issue No. 238). “In the case of cancer treatment, we’re most likely going to go down a similar path.”
 LÉO RAMOS CHAVESTwo recent studies by his group at InCor published in 2014 and 2016 in The American Journal of Physiology – Heart and Circulatory Physiology showed that physical exercise in people with heart problems can deactivate protein degradation processes in muscle cells, stimulate the production of anti-inflammatory cytokines, and improve the flow of calcium. This is essential for proper muscle functioning, especially in the case of the heart, the functioning of which may be hindered by anti-tumor drugs or cachexia. In an article published in May in Oncology Reports, researchers in France argue that “physical exercise appears as an interesting non-pharmaceutical way to counteract cancer cachexia-induced-heart failure.” They found that aerobic training has anti-inflammatory effects and prevents heart muscle atrophy.
LÉO RAMOS CHAVESTwo recent studies by his group at InCor published in 2014 and 2016 in The American Journal of Physiology – Heart and Circulatory Physiology showed that physical exercise in people with heart problems can deactivate protein degradation processes in muscle cells, stimulate the production of anti-inflammatory cytokines, and improve the flow of calcium. This is essential for proper muscle functioning, especially in the case of the heart, the functioning of which may be hindered by anti-tumor drugs or cachexia. In an article published in May in Oncology Reports, researchers in France argue that “physical exercise appears as an interesting non-pharmaceutical way to counteract cancer cachexia-induced-heart failure.” They found that aerobic training has anti-inflammatory effects and prevents heart muscle atrophy.
Research projects
1. Systemic Inflammation in patients with Cachexia Associated with Cancer: Mechanisms and therapeutic strategies within a translation medicine-based approach (No. 12/50079-0); Grant Mechanism Thematic Project; Principal Investigator Marília Cerqueira Leite Seelander (USP); Investment R$2,246,952.23.
2. Molecular Basis of Cachexia: Adipogenesis and extracellular matrix remodeling of white adipose tissue in patients with gastrointestinal cancer (No. 10/51078-1); Grant Mechanism Junior Researcher; Principal Investigator Miguel Luiz Batista Jr. (UMC); Investment R$910,407.63.
Scientific articles
ANTUNES-CORREA, LM et al. Molecular basis for the improvement in muscle metaboreflex and mechanoreflex control in exercise-trained humans with chronic heart failure. American Journal of Physiology. V. 307, No. 11, p. 1655–66. 2014.
BATISTA, M. L. Jr., Cachexia-associated adipose tissue morphological rearrangement in gastrointestinal cancer patients. Journal of Cachexia, Sarcopenia and Muscle. V. 7, No. 1, p. 37–47. 2016.
BELLOUM, Y. et al. Cancer-induced cardiac cachexia: Pathogenesis and impact of physical activity. Oncology Reports. V. 37, No. 5, p. 2543–52. 2017.
BELUZI, M. et al. Pioglitazone treatment increases survival and prevents body weight loss in tumor-bearing animals: Possible anti-cachectic effect. PLoS One. V. 10, No. 3, p. 1–16. 2015.
LIRA, F.S. et al. The therapeutic potential of exercise to treat cachexia. Current Opinion in Supportive and Palliative Care. V. 9, No. 4, p. 317–24. 2015.
MOORE, S. C. et al. Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA Internal Medicine. V. 176, No. 6, p. 816–25. 2016.
NOBRE, T. S. et al. Exercise training improves neurovascular control and calcium cycling gene expression in patients with heart failure with cardiac resynchronization therapy. American Journal of Physiology. V. 311, No. 5, p. 1180–88. 2016.
VAN DE BOOL, C. et al. A randomized clinical trial investigating the efficacy of targeted nutrition as adjunct to exercise training in COPD. Journal of Cachexia, Sarcopenia and Muscle. 2017 (in press).