
Science Photo Library
Illustration of fetus during pregnancyScience Photo LibraryTwo recent studies by research teams whose members include Brazilian scientists have provided more evidence that there is a window of time during which Zika virus infection in pregnant women can severely damage the fetus, triggering microcephaly in the most serious cases. Once this critical phase has passed—roughly equivalent to the first trimester of pregnancy—the risk drops substantially for the children of women contaminated by the virus, although it cannot be affirmed that later infections do no harm at all.
In a paper published in the journal PNAS on February 13, 2017, an international team—which includes Brazilian biochemist Sergio Verjovski-Almeida of the Butantan Institute and the University of São Paulo Chemistry Institute (IQ-USP)—stated that the human placenta is more sensitive to infection from the virus during the first three months of pregnancy. During this phase, the placenta, which carries the genetic material of the fetus, has not yet acquired all of its immune defenses, while it also produces proteins that stimulate the infectious agent to attach to and enter the cells of the developing baby.
Another study, led by physician José Xavier-Neto, of the Brazilian Biosciences National Laboratory (LNBio), in Campinas, has indicated that the Zika infection only causes serious abnormalities in mouse pups when their mothers are exposed to the pathogen between the fifth and twelfth day following fertilization. In humans, this window corresponds to the period from the second to fifth week of pregnancy. The Zika infection caused no significant malformations to the rodent offspring when it occurred following the twelfth day after mating. The paper was released in the online edition of PLOS Neglected Tropical Diseases on February 23, 2017.
Mature, resistant placenta
The first study sought to find an explanation for the evidence that the fetuses of women infected with Zika during the first three months of pregnancy have a greater risk of presenting health issues like microcephaly at birth than the babies of mothers who have contact with the pathogen in later stages of pregnancy. According to the PNAS article, the mature placenta, at the end of pregnancy, is much more resistant to the viral infection than the primitive placenta of the first three months. In this early stage of pregnancy, the organ that communicates between mother and fetus produces proteins called attachment receptors that stimulate the infectious agent to attach to and enter the cells of the developing baby. According to the authors, these cells have not fully formed the defense system needed to block entry of the Zika virus.
As time goes by, the more mature placenta creates a completely different environment, unfavorable to the advance of the maternal virus into the fetus. “At the end of pregnancy, the placental cells are resistant to infection by the virus because they don’t express the genes that encode the proteins responsible for promoting viral attachment and entry into the fetal tissue although, at the same time, they activate a number of genes associated with antiviral defense,” explains Verjovski-Almeida.
In 2016, Russian biologist Dinar Yunusov, then Verjovski-Almeida’s doctoral student at the IQ, performed gene expression analyses both on cells in the mature placenta and on an experimental model composed of reprogrammed embryonic stem cells, mimicking a first-trimester placenta. “Zika really seems to need the presence of these attachment receptors to attack fetal cells,” says Yunusov, who is currently doing postdoctoral work in the United States.
Given this sign that the placenta is more vulnerable to Zika during the first trimester of pregnancy, Verjovski-Almeida says that it might be helpful to develop some way to reinforce the organ’s biological defenses, like developing a serum that can be administered to women infected by the virus in early pregnancy. “But we have to research this cautiously, because it’s very tricky to prescribe something to women in the early months of pregnancy,” the biochemist says. In parallel with this line of research, Verjovski-Almeida plans to verify whether the pattern of activation for the genes that control the production of viral receptors in the early placenta can vary from individual to individual within a population and be used as a test to signal the organ’s greater or lesser vulnerability to attack by the Zika virus.
A study published in the Journal of the American Medical Association in late 2016 indicated that 5.9% of babies born to 442 U.S. women infected by the Zika virus displayed microcephaly or other brain abnormalities, while no anomalies were found in those born to women who had contact with the virus during their second or third trimester. “There is evidence that the early placenta is also permissive to other viruses, such as rubella,” says researcher R. Michael Roberts of the University of Missouri and leader of the team that conducted the Zika experiments with mature and early placentas, published in PNAS.
Another interesting piece of data from the same article involves the evolution of the pathogen. In laboratory experiments, the original Zika strain, which came from Uganda, infected the early placenta model faster and did more damage than the Asian strain, which reached the Americas and resembles the one that circulates in Brazil and causes microcephaly. The researchers concluded that the African variety is so aggressive that it can destroy the placenta during the first trimester. This means that infection by the African strain of Zika in early pregnancy could effectively terminate the pregnancy. “The aggressive nature of the African strain may explain why no cases of microcephaly have been recorded in Africa,” Verjovski-Almeida conjectures. “Infected fetuses would not even be born. They would be miscarried first.”
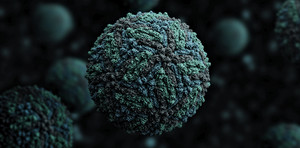
Manuel Almagro Rivas / Wikimedia Commons
3-dimensional representation of the Zika virus, which crosses the placenta and damages brain tissueManuel Almagro Rivas / Wikimedia CommonsHydrocephaly and microcephaly
The work of the LNBio team entailed development of an animal model of Zika infection that resembles the infection in humans. In two lines of wild-type immunocompetent mice – meaning that their immune systems were able to fight off infections – researchers injected the virus into the jugular vein of pregnant females at different stages of gestation, from 5.5 to 19.5 days after fertilization. This allowed them to observe the sequence of problems that the virus causes in mouse offspring as a function of the stage of pregnancy when Zika exposure took place. “We wanted to map the critical window during which infection in pregnancy causes congenital abnormalities,” explains José Xavier-Neto.
The pups of female mice contaminated five days after fertilization presented a series of developmental problems: incomplete closure of the neural tube (dysraphia); hydrocephaly (accumulation of cerebrospinal fluid in the brain, which causes the brain to swell and enlarges the cranium); delayed growth of the embryo; and other severe malformations. “In humans, hydrocephaly is a harbinger of microcephaly,” says Xavier-Neto. When the infection occurred from 7.5 to 9.5 days after fertilization, the clinical presentation of the pups was not limited to microcephaly. They also displayed intra-amniotic hemorrhage, widespread edema, and little vascularization, especially in the cephalic region. Some embryos even died in the uterus following infection. According to the researcher, the animal model of Zika developed at LNBio is the only one to show dysraphia, hydrocephaly, and arthrogryposis (congenital contracture of the joints, which leads to the formation of twisted or bent hands and feet).
Infections induced following the 12th day after fertilization did not cause major damage in the mouse fetuses. Although the Zika genome was not found in the brain tissue of embryos in this stage of gestation, it was detected in cells of the spleen, liver, and kidneys of developing mice. “We can’t say that there is a safe period of pregnancy for Zika infection to take place,” explains cardiologist Kleber Franchini, with the LNBio, and another author of the study.
There is a reasonable amount of research indicating that the appearance of the congenital anomalies associated with Zika depends on the timing of the infection during pregnancy. Some studies also suggest that other factors, like the virulence of the viral strain and a population’s genetic specificities, also favor more or less serious outcomes in Zika infection. In an article published in the journal Nature on May 11, 2016, researchers at the University of California, San Diego (UCSD), and USP reported on an experiment that illustrates this situation.
Between the 10th and 12th days of gestation, the scientists at UCSD and USP injected Zika into the tails of two lineages of mice, one of which differed from the variety of wild-type mice used in the LNBio experiment. One lineage presented a series of malformations associated with infection by the virus, including microcephaly. The other produced pups that were resistant to the infection. “The timing of Zika infection is undoubtedly important in determining the severity of the damage, but the immune response of each individual also appears to be crucial,” says neuroimmunologist Jean Pierre Peron of the Institute of Biomedical Sciences (ICB) at USP and one of the authors of the study published in 2016. “The most interesting part of the LNBio team’s study was that it showed the occurrence of arthrogryposis in an animal model, something that hadn’t been observed yet.”
 Outbreak prompts upsurge in scientific papers
Outbreak prompts upsurge in scientific papers
The number of articles on the Zika virus increased 20-fold in two years; Brazil ranks second in the output of studies on the disease
A bibliometric survey by a team from the Oswaldo Cruz Foundation (Fiocruz) and the Federal University of Rio de Janeiro (UFRJ) indicates that the production of scientific articles on the Zika virus rose about 20-fold following confirmation that the pathogen was associated with cases of microcephaly in Brazil and after the disease became an international health concern. Authors whose professional addresses are in Brazil produced more papers on the virus, second only to U.S. researchers and outranking the British. The article was published in the journal PLOS Neglected Tropical Diseases on January 19, 2017.
From 1952, when the virus was first isolated in Uganda, until 2013, the year of the Zika outbreak in French Polynesia, researchers released 44 documents in the Scopus database and 28 in the Web of Science that contained the expression “Zika virus” in the title. From January 2014 to August 2016, the number of similar articles in the two databases totaled about 1,300 (see graph). A comparison of scientific output indexed by the search engine Google Academics and containing the words “Zika virus” found similar figures: 47 papers from 1952 to 2013 and about 1,600 from 2014 to 2016.
In both Scopus and Web of Science, most of the scientific articles on Zika were written by researchers from 18 countries. U.S. scientists accounted for about 31% of the papers, while Brazilians were responsible for 12%. British scientists came in third, authoring 9% of the studies. “We need to recognize that the efficiency of Brazilian scientists has been truly commendable, despite their countless logistical and funding challenges,” observes pharmacist Marcio L. Rodrigues, vice-coordinator general of the Center for Technological Development in Health (CDTS) at Fiocruz, associate professor at the UFRJ Microbiology Institute, and co-author of the study, alongside Priscila Albuquerque. Basic research is still the focus of the scientific drive in the realm of Zika. The survey found no significant change in the number of patent applications related to the disease worldwide.
Projects
1. Characterization of the mechanisms of action of long non-coding RNAs involved with gene activation programs in human cells (nº 14/03620-2); Grant Mechanism Thematic project; Principal Investigator Sergio Verjovski-Almeida (USP); Investment R$2,149,830.50.
2. Functional characterization of long non-coding RNA transcribed from the antisense strand in the VEGFA gene locus (nº 10/51152-7); Grant Mechanism Scholarships in Brazil – Doctorate (Direct); Principal Investigator Sergio Verjovski-Almeida (USP); Grant Recipient: Dinar Yunusov; Investment: R$212,228.31.
3. The evolutionary origins of regulatory networks that segment the heart into inflow and outflow chambers (nº 13/22695-0); Grant Mechanism Regular Research Grant; Principal Investigator José Xavier-Neto (LNBio); Investment R$690,098.30.
Scientific articles
SHERIDAN, M. A. et al. Vulnerability of primitive human placental trophoblast to Zika virus. PNAS. February 13, 2016.
XAVIER-NETO, J. et al. Hydrocephalus and arthrogryposis in an immunocompetent mouse model of ZIKA teratogeny: A developmental study. PLOS Neglected Tropical Diseases. February 23, 2017.

