
Isadora Clivatti Furigo /USP
Neurônios da fome (círculos vermelhos) ativados pelo hormônio do crescimento durante o jejumIsadora Clivatti Furigo /USPTodos que já fizeram dieta para emagrecer conhecem o processo. Vencida a inércia e iniciado o regime, os dígitos da balança no começo costumam ceder, seja qual for a estratégia adotada. O difícil é manter o ritmo de emagrecimento. Mesmo com a ajuda de medicamentos, em poucas semanas o peso estaciona ou, pior, volta a subir. A forma como o organismo dos seres humanos e de outros animais foi moldado ao longo da evolução explica, em parte, a dificuldade de emagrecer. Milhares de anos de pouca disponibilidade de alimentos (cuja obtenção exigia esforço físico) muniram o corpo de potentes e complexas estratégias de economizar energia. Em experimentos com camundongos, o grupo coordenado pelo fisiologista José Donato Junior, do Instituto de Ciências Biomédicas da Universidade de São Paulo (ICB-USP), descreveu mais um mecanismo que atua contra a perda de peso.
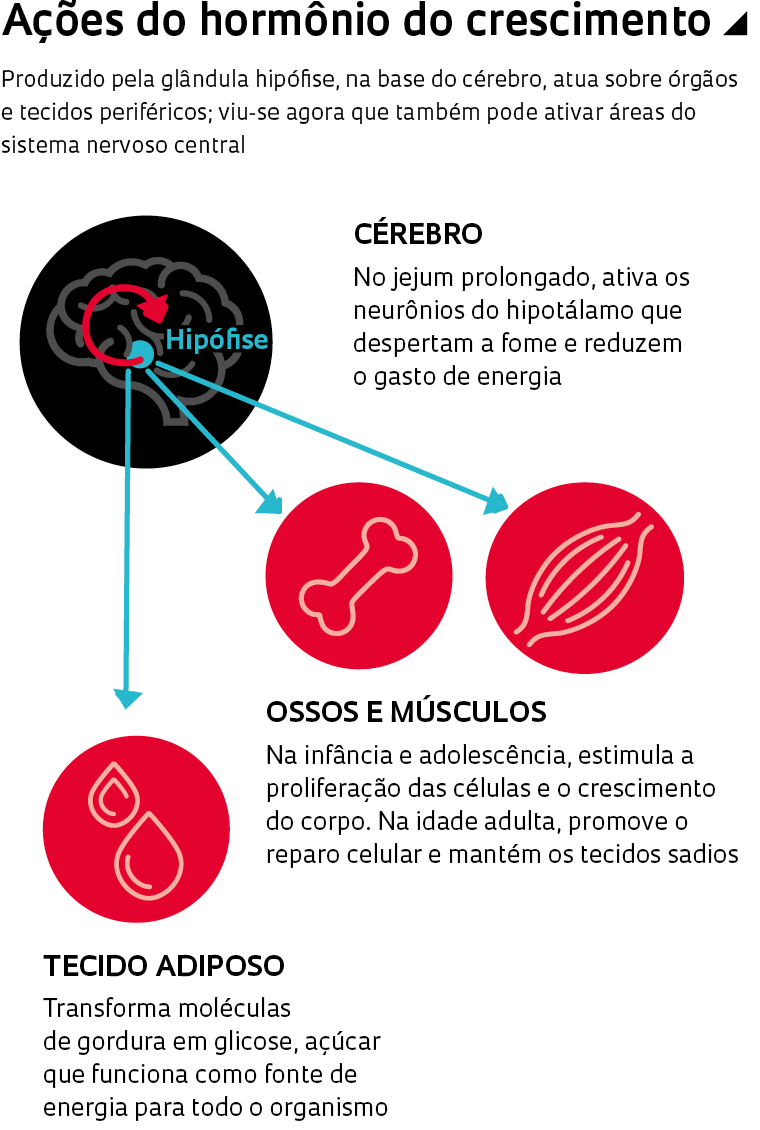
Seu orquestrador é um composto até então insuspeito: o hormônio do crescimento. Conhecido há quase um século e produzido pela hipófise, uma glândula na base do cérebro, o hormônio do crescimento age sobre os tecidos periféricos. Secretado em maior quantidade durante o sono, aumenta o gasto de calorias, liberando a energia armazenada no tecido adiposo e induzindo a multiplicação e o reparo das células em ossos, músculos e vísceras. Agora, a equipe da USP verificou que, quando se come menos que o habitual, esse hormônio funciona de modo diferente. Ele ativa uma região do cérebro que aumenta a fome e reduz o gasto de energia. Como consequência, fica mais difícil emagrecer.
Donato e colaboradores apresentaram evidências desse efeito, nunca antes notado, em experimentos com camundongos descritos em um artigo na edição de 8 de fevereiro da revista Nature Communications. “Ao restringir a ingestão de calorias, o nível desse hormônio sobe e induz o corpo a entrar em modo de a sobrevivência: ele deixa de influenciar o crescimento e indica que é preciso comer e guardar energia”, explica o fisiologista.
O achado, de certo modo, surpreende especialistas na área. “Geralmente se identificam funções novas para hormônios já conhecidos, mas nenhuma tão importante quanto essa”, afirma o endocrinologista Lício Velloso, da Universidade Estadual de Campinas (Unicamp) e estudioso dos mecanismos bioquímicos associados à obesidade e ao diabetes. “Nunca havia ficado tão evidente que, no cérebro, esse hormônio pudesse regular a fome e o gasto energético.”
Desde os anos 1960 se sabe que o jejum aumenta a liberação de hormônio do crescimento. Em testes com voluntários saudáveis em um hospital de Nova York, o médico Solomon Berson (1918-1972) e a física-médica Rosalyn Yalow (1921-2011) observaram que a privação prolongada de nutrientes (dois dias e meio sem comer) aumentava a liberação de hormônio do crescimento. Não se sabia, porém, qual era a consequência disso nem por que acontecia. “Parecia ser um efeito paradoxal”, conta Donato. “Por que o organismo em carência de nutrientes iria direcionar seus esforços para crescer, o que consome mais energia?”

A resposta veio com os resultados publicados em fevereiro. Interessado em conhecer como os hormônios atuam no sistema nervoso central, Donato e sua equipe olharam para o cérebro, em vez de mirar os tecidos periféricos, como fazem outros grupos. “A ação do hormônio do crescimento sobre os centros reguladores da fome vinha sendo negligenciada”, comenta o bioquímico brasileiro Marcelo Dietrich, da Escola de Medicina da Universidade Yale, Estados Unidos, que estuda os mecanismos bioquímicos controladores da fome e da saciedade.
O primeiro passo foi descobrir onde o hormônio do crescimento agia no cérebro e seu efeito sobre a fome e o consumo de energia. Em um teste inicial, a fisiologista Isadora Clivatti Furigo, em seu pós-doutorado sob a supervisão de Donato, administrou a roedores saudáveis hormônio do crescimento marcado com um material que permite o rastreamento. Em seguida, examinou o hipotálamo, pequena região do cérebro associada ao controle da temperatura corporal, da sede, do cansaço, do sono e, claro, da fome. Ela observou que o hormônio se ligava às células de ao menos 10 áreas. Uma em especial chamou a atenção: o núcleo arqueado. Essa área diminuta contém um grupo pequeno de células (alguns milhares no camundongo) sabidamente associadas à regulação da ingestão de alimentos e do gasto energético. Apelidadas de neurônios da fome, essas células produzem dois compostos – o neuropeptídeo conhecido pela sigla AgRP e o neuropeptídeo Y (NPY) – razão por que são chamados de neurônios AgRP/NPY – que agem em outras regiões cerebrais e levam o corpo a buscar comida.
Um gatilho da fome
O aumento da concentração do hormônio do crescimento no hipotálamo, de fato, despertou os neurônios da fome. Um dia após receber uma dose do hormônio, os roedores passaram a comer mais do que os camundongos tratados com uma solução inócua (placebo). Os animais do primeiro grupo também gastavam menos energia do que os do segundo, sugerindo que níveis elevados de hormônio do crescimento no cérebro funcionavam como um gatilho da fome e da economia de energia. “O trabalho atual abre um novo campo de investigação”, afirma o endocrinologista Cesar Boguszewski, coordenador da Unidade de Neuroendocrinologia do Hospital de Clínicas da Universidade Federal do Paraná (UFPR). “Caso esse efeito seja observado em seres humanos, pode ajudar a entender por que as dietas de emagrecimento muitas vezes falham.”
Para confirmar que o aumento da fome era decorrente da ação desse hormônio sobre os neurônios AgRP/NPY, os pesquisadores decidiram torná-los insensíveis ao hormônio do crescimento. Donato e Furigo receberam camundongos geneticamente alterados para não apresentar o receptor do hormônio em todo o cérebro. Eles foram preparados pelo biólogo molecular norte-americano John Kopchick, da Universidade de Ohio, Estados Unidos, desenvolvedor de um medicamento que impede a ação do hormônio do crescimento. No Laboratório de Neuroanatomia Funcional da USP, a dupla brasileira restringiu a ausência do receptor só aos neurônios da fome usando uma estratégia de manipulação genética chamada recombinação Cre-Lox – anterior à Crispr, hoje em moda.
Tratados com a dieta padrão dos laboratórios, os camundongos sem o receptor do hormônio nos neurônios AgRP/NPY comiam o mesmo que os animais sem alteração genética. Também apresentavam nível de atividade, peso e composição corporal (massa gorda) semelhantes. Submetidos por cinco dias a uma dieta na qual comiam 40% da quantidade normal de ração, os animais sem alteração genética passaram a se movimentar menos e a economizar energia. Já os camundongos com neurônios da fome sem receptor do hormônio do crescimento quase não apresentaram mudanças no nível de atividade. Nessa situação, este grupo emagreceu mais: perdeu 20% mais peso, principalmente massa gorda, do que os animais do primeiro grupo. “Sem o receptor para hormônio do crescimento, os neurônios AgRP/NPY parecem não perceber adequadamente a privação de alimentos”, conta Furigo.
Na fase seguinte, os pesquisadores testaram algo mais próximo de um possível tratamento de perda de peso para seres humanos. Restringiram a quantidade de alimento e deram aos roedores duas doses diárias de pegvisomanto, medicamento usado para tratar pessoas com acromegalia – aumento exagerado das extremidades do corpo pela produção excessiva de hormônio do crescimento na idade adulta. Assim, tentavam maximizar a perda de peso sem mexer nos genes. Com os roedores, funcionou. Os camundongos que receberam pegvisomanto emagreceram mais do que os do grupo tratado com placebo porque mantiveram gasto energético elevado mesmo passando fome. “Esse resultado é uma prova de conceito de que talvez seja possível usar esse tipo de medicamento para auxiliar as pessoas a perderem mais peso”, sugere Donato. “Em uma dieta, um tratamento com o composto talvez faça os níveis de hormônio do crescimento voltarem ao normal e, assim, reduzir a fome e manter o gasto de energia.”
Alternativa desejável
Os pesquisadores suspeitam que o efeito observado em camundongos também ocorra em outros animais e nos seres humanos. “Mecanismos fundamentais para a sobrevivência costumam ser preservados ao longo da evolução”, lembra Dietrich, de Yale. No entanto, a confirmação dessa ação benéfica e a verificação de eventuais efeitos colaterais exigirão muitos experimentos. Se funcionar como o previsto, seria uma alternativa bem-vinda para auxiliar o tratamento da obesidade. Há urgência em encontrar formas efetivas de controlar o excesso de peso, problema que atinge 1,9 bilhão de pessoas no mundo e, no caso humano, tem causas múltiplas (genéticas, ambientais e comportamentais).
Ainda que o bloqueio do hormônio favoreça o emagrecimento humano, Boguszewski, da UFPR, prevê uma dificuldade para adotar a estratégia na prática clínica. “Primeiro, seria necessário limitar o efeito a uma área restrita do cérebro”, diz o pesquisador, que investiga ação periférica do hormônio do crescimento. Quase todos os tecidos do corpo têm receptores para ele, e impedir sua ação pode trazer efeitos graves. “Ainda estamos na fase de tentar compreender melhor o funcionamento do hipotálamo, antes de pensar em estratégias terapêuticas mais eficientes”, adverte Velloso. “Do ponto de vista biológico, somos protegidos para não morrer de desnutrição.”
Logo após sua descoberta em 1994, a leptina se tornou conhecida como o hormônio da saciedade. Produzida pelo tecido adiposo, ela sinaliza para o cérebro se o estoque de energia armazenado na forma de gordura está adequado ou se é preciso comer. Essa, porém, não é sua única função. No início da vida, ela é importante para o desenvolvimento do cérebro e dos sistemas imunológico e reprodutivo. Faltava saber se os efeitos de sua carência poderiam ser revertidos.
“Alguns, sim; outros, apenas parcialmente”, afirma o fisiologista José Donato Junior, da USP. A fisiologista Angela Ramos Lobo e Donato, seu orientador no doutorado, elaboraram uma estratégia sofisticada para verificar essa possibilidade. Desativaram em roedores o gene que codifica o receptor da leptina. Os camundongos produziam a leptina, mas o corpo não percebia seu efeito.
Os pesquisadores reativaram esse gene na quarta semana de vida de um grupo de animais e na décima de outro – idades que correspondem ao final da infância e da adolescência em seres humanos – e os acompanharam por mais seis meses.

Léo Ramos Chaves
Irmãos e diferentes: o camundongo obeso (à esq.) não produz o receptor da leptinaLéo Ramos ChavesAssim como os roedores que não produzem leptina, os animais privados de sua ação no início da vida comiam demais, gastavam pouca energia e eram obesos e inférteis. Também tinham o cérebro 10% menor do que os do grupo de controle.
Restabelecida a ação da leptina, os animais passaram a comer menos, emagreceram e recuperaram em parte o desenvolvimento cerebral. Ainda assim, continuavam obesos, inférteis e com maior propensão a ter diabetes, segundo artigo publicado em 29 de janeiro na revista eLife. “A sinalização da leptina no início do desenvolvimento define o padrão de gasto energético”, explica Donato.
Suspeita-se que esses achados possam ser aplicados a seres humanos. Embora a deficiência de leptina seja rara (há uns 30 casos conhecidos no mundo), é comum a sinalização disfuncional, decorrente de carência nutricional ou do excesso de peso na infância – nesses casos, o corpo produz o hormônio, mas não o reconhece. “Nessa situação, é importante agir cedo, com reposição ou mudança de hábitos alimentares, para garantir o melhor desenvolvimento possível”, afirma Lício Velloso, da Unicamp.
Projetos
1. Ação do hormônio do crescimento no sistema nervoso: Relevância para as funções neurais e na doença (nº 17/02983-2); Modalidade Projeto Temático; Pesquisador responsável José Donato Junior (ICB-USP); Investimento R$ 1.238.502,38.
2. Estudo dos efeitos centrais do hormônio do crescimento sobre o metabolismo energético e controle glicêmico (nº 16/09679-4); Modalidade Bolsa no País – Pós-doutorado; Pesquisador responsável José Donato Junior (ICB-USP); Bolsista Isadora Clivatti Furigo; Investimento R$ 296.549,92.
Artigo científico
FURIGO, I. C. et al. Growth hormone regulates neuroendocrine responses to weight loss via AgRP neurons. Nature Communications. 8 fev. 2019.