The first cases of acute lung damage linked to the use of electronic cigarettes started to show up in the USA in April 2019. It was a surprise for the doctors. Young adults around 20 years of age, a portion of whom had no previous lung problems, arrived in US hospitals suffering from shortness of breath, coughs, and chest pain, often associated with abdominal pain, nausea, vomiting, diarrhea, fatigue, fever, and weight loss. Among the different symptoms was a common theme: they were all vape users. The symptoms had been developing over the previous weeks and, in some cases, in just a few days. Some of these patients had to be transferred to Intensive Care Units; in August that year, the first deaths were reported.
The US Centers for Disease Control and Prevention (CDC) then started monitoring the occurrence of this new disease, which was named EVALI (E-cigarette or Vaping product use-Associated Lung Injury). For now, only one major outbreak of EVALI has been verified. According to CDC data, between the start of 2019 and February 18, 2020, 2,807 hospitalizations and 68 deaths were attributed to the disease in the 50 US states and territories. Over half of the deaths occurred in obese patients or those with some prior health issue. In February 2020, the agency stopped collecting data from the states due to the decreased incidence of cases and the outbreak of the Covid-19 pandemic. The symptoms and lung damage of EVALI are similar to those of Covid-19.
In the UK, another large market for electronic cigarettes, three deaths and 231 records of adverse reactions associated with the use of these devices were reported between May 2016 and January 2021. The information comes from an article published in the British newspaper The Observer in August this year.
In Brazil, the Brazilian Health Regulatory Agency (ANVISA) has counted seven cases of EVALI. For physicians, this number certainly does not reflect reality, considering that notification of the disease is not compulsory in the country. A request by the Brazilian Society of Pulmonology and Phthisiology (SBPT) for the compulsory notification of EVALI was not granted by the agency. “ANVISA basically responded that notification is not compulsory because EVALI is a disease that affects users of a product whose sale is prohibited,” explains pulmonologist Paulo Corrêa, coordinator of the SBPT Scientific Commission on Smoking.
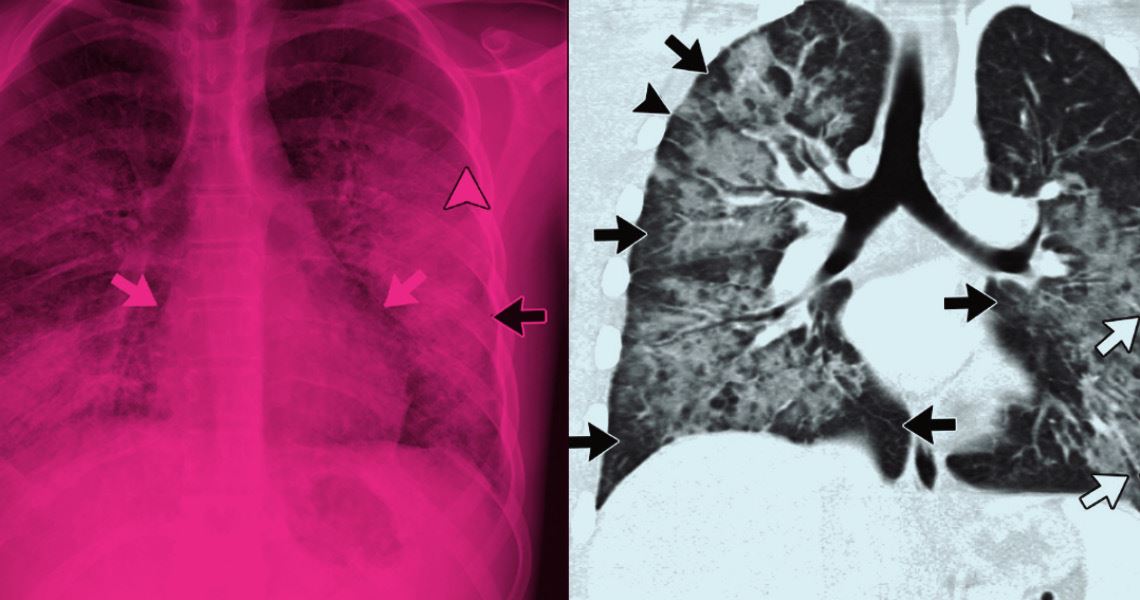
There is no specific test or examination to detect EVALI. Diagnosis is made by exclusion of other lung diseases and by analysis of the patient’s history of using electronic cigarettes. A suspected case of the disease was attended by pulmonologist Felipe Marques, of the Hospital Beneficência Portuguesa of São Paulo. The occurrence was described in an article published in the May/June 2021 edition of the Jornal Brasileiro de Pneumologia.
According to Marques, the patient was a 48-year-old woman with a persistent cough and fatigue. She had been treated with antibiotics, but her condition did not improve. Blood and RT-PCR tests were done to check if the patient had been infected with the Sars-CoV-2 virus, but the results were negative. However, when she underwent a computerized tomography of the chest, she showed characteristic symptoms of viral pneumonia caused by Covid-19. “She presented that pattern of opacity that we call ground-glass,” explains Marques.
According to the pulmonologist, the patient was an ex-smoker for 21 years, but had been using electronic cigarettes for the entirety of the first half of 2020. “We treated the condition with anti-inflammatories and asked her to stop using electronic cigarettes; she began to improve in the first days,” he says. One month later, the tomography showed almost full recovery from the damage.
The causes of acute conditions related to vaping, like this one treated in Brazil, are still being studied. Another scientific article published in the periodical The New England Journal of Medicine in February 2020 offered an important clue: the presence of Vitamin E acetate in the lung fluid of 48 patients from a sample of 51 individuals diagnosed with EVALI. In a control group, composed of 99 healthy individuals, including 18 electronic cigarette smokers, the substance was not found. The study was coordinated by CDC researchers.
Vitamin E acetate is a viscous substance used as an additive in electronic cigarettes, especially those that contain the cannabinoid THC, derived from plants of the Cannabis genus. Its presence can also be found in vitamin supplements and dermatological products.
“The hypothesis that vitamin E acetate causes the disease still needs to be validated,” says Marques. “There are many other components of electronic cigarettes that could be pernicious when inhaled.”
Another harmful substance identified in the liquids of electronic cigarettes is the flavoring diacetyl. It is also related to an acute lung disease: bronchiolitis obliterans. Also known as “popcorn lung,” bronchiolitis obliterans has been documented in workers of a company that manufactured microwave popcorn. A study published in 2019 in the Canadian Medical Association Journal identified this disease in a 17-year-old vape smoker. Previously, in 2015, a study that was published in the US journal Chest Infections had already diagnosed bronchiolitis obliterans in a 60-year-old electronic cigarette smoker.
Scientific article
MEDEIROS, A. K. et al. Diagnóstico diferencial entre lesão pulmonar associada ao uso de cigarro eletrônico e pneumonia por Covid-19. Jornal Brasileiro de Pneumologia. vol. 47, no. 3. 2021.
BLOUNT, B. C. et al. Vitamin E acetate in bronchoalveolar-lavage fluid associated with Evali. New England Journal of Medicine. feb. 20, 2020.
LANDMAN, S. T. et al. Life-threatening bronchiolitis related to electronic cigarette use in a Canadian youth. Canadian Medical Association Journal. nov. 20, 2019.
ATKINS, G. et al. Acute inhalational lung injury related to the use of electronic nicotine delivery system (ENDS). Chest Infections. vol. 148, no. 4. oct. 1, 2015.