 MECALEHA/GETTY IMAGESAdvertisements by fast-food chains commonly feature super-size portions—double hamburgers accompanied by large servings of French fries and a huge soda to complete the meal. But this mountain of calories, often well above the recommended amount for a meal, does not always appease hunger. Actually, the higher the fat content of the food, the more people want to eat. For those who expect otherwise, that these heavier and hard-to-digest foods should more easily result in satiety, there is now an explanation. “A high-fat diet makes the neurons that induce hunger more active,” says Marcelo Dietrich, biochemist and researcher at Yale University School of Medicine.
MECALEHA/GETTY IMAGESAdvertisements by fast-food chains commonly feature super-size portions—double hamburgers accompanied by large servings of French fries and a huge soda to complete the meal. But this mountain of calories, often well above the recommended amount for a meal, does not always appease hunger. Actually, the higher the fat content of the food, the more people want to eat. For those who expect otherwise, that these heavier and hard-to-digest foods should more easily result in satiety, there is now an explanation. “A high-fat diet makes the neurons that induce hunger more active,” says Marcelo Dietrich, biochemist and researcher at Yale University School of Medicine.
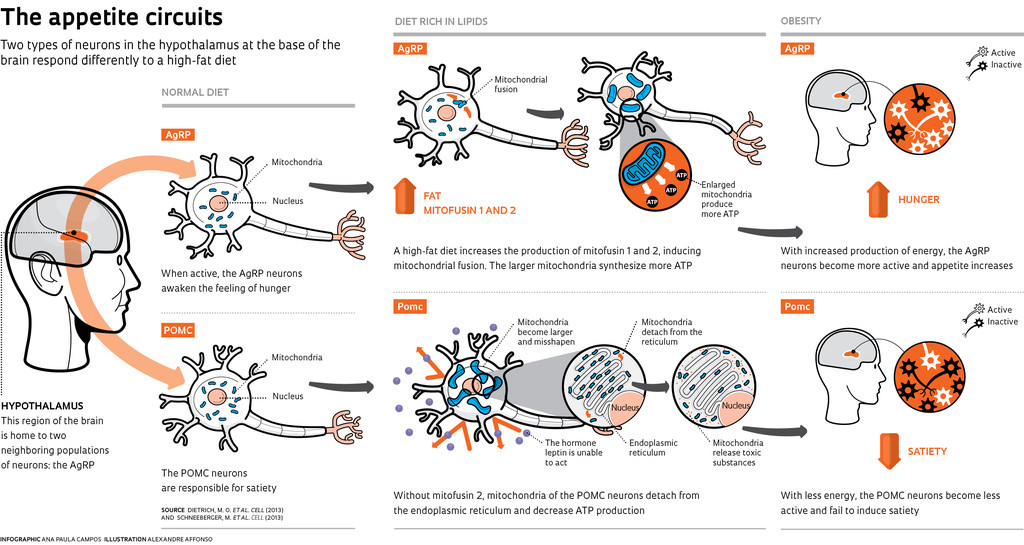
He recently reached this conclusion by studying the action of two groups of neurons—one that induces hunger and another that induces satiety—both located in a region at the base of the brain called the hypothalamus. In experiments with mice, Dietrich found that consuming a lot of fat deregulates this mechanism, which is essential to survival. By feeding mice different types of foods, he found that excess fat increases the activity of hunger neurons, known by the acronym AgRP, while it reduces the functioning of the satiety neurons, the POMC. This imbalance arises as a result of changes in the mitochondria, the organelles that produce energy in neurons, as Dietrich showed in a study published in September 2013 in the journal Cell. In addition to revealing the origin of the voracious appetite for fatty foods, these findings underscore the difficulty in developing drugs against obesity that are based on the modulation of these two types of neurons. This is because a single protein induces distinct changes in the mitochondria of these cells.
Relentless hunger
The focus of the studies by Dietrich, who is also associated with the Federal University of Rio Grande do Sul (UFRGS) Department of Biochemistry, where he works with biochemist Diogo Souza, is the AgRP neurons, which when activated awaken appetite. “It is a mechanism that is essential to life,” he says. “Even during fasting, when energy is scarce and the metabolism of many cells decreases, a portion of these neurons continues to consume energy to ensure the urge to find food,” he explains. In an experiment conducted with mice in the laboratory of Tamas Horvath, director of the Yale Program on Integrative Cell Signaling and Neurobiology of Metabolism, Dietrich and his colleagues wanted to know what would happen if mice were given food fortified with fat (lipids). They observed that after the meal, the proportion of these neurons still active in the brains of the mice that consumed fat was higher than in the mice on normal diets. This result was the opposite of what was expected, since the first group of mice had consumed more energy.
The researchers showed that this abnormal activation occurs because the mitochondria, producers of cellular energy, adenosine triphosphate (ATP), fuse. Larger and less abundant, mitochondria generate more ATP and boost the activity of AgRP neurons, increasing hunger and fat accumulation. This results in mice that are well above normal weight.
“Mitochondrial fusion caused by a high-fat diet is a new discovery,” says Dietrich. Conversely, when the body is in starvation mode, mitochondria divide. Smaller and more abundant, they are less efficient in the production of ATP. He proved the effect that fats have on mitochondria by feeding mice a high-fat diet, genetically engineered to produce two proteins: mitofusin 1 and mitofusin 2, which are responsible for the fusion of these organelles. By blocking the fusion of mitochondria—they retain their normal size—a larger proportion of hunger neurons remained at rest, and the mice did not become fat.
Insatiable
Dietrich’s findings are even more significant when viewed in conjunction with another article that appeared in the same issue of Cell. In this second paper, to which Dietrich and Horvath contributed, researchers Marc Schneeberger and Marc Claret of the Barcelona-based August Pi i Sunyer Biomedical Research Institute (IDIBPS) identified in mice another relationship between diet and how the POMC neurons responsible for satiety work. The absence of mitofusin 2, whose production drops when mice are fed a diet rich in lipids, nearly sabotages those brain cells. “They become less active,” says Dietrich. “Since the POMC neurons promote satiety, their inactivation disrupts the equilibrium, and only the hunger neurons remain in high activity. “With unchecked appetites, mice become extremely obese.”
 INFOGRAPHIC: ANA PAULA CAMPOS ILLUSTRATION: ALEXANDRE AFFONSO
INFOGRAPHIC: ANA PAULA CAMPOS ILLUSTRATION: ALEXANDRE AFFONSO
The problem behind the malfunctioning POMC is again in the mitochondria, which, this time, become larger and misshapen. Without mitofusin 2, mitochondria, in addition to being deformed, detach from the endoplasmic reticulum, the organelle involved in protein synthesis. “We believe that mitochondria use calcium and lipids stored in the reticulum to generate energy,” says Dietrich. When this chain of events is interrupted, both are damaged and malfunction. In this context, the mitochondria begin to release reactive types of oxygen, molecules that cause biochemical imbalances in the body. Under these conditions, the mitochondria fail to produce the necessary ATP for the POMC neurons to function, and while they are inactive, these neurons stop responding to leptin, the hormone responsible for signaling the body to feel sated. Satiety does not occur, and the gluttonous mice become efficient at accumulating fat.
Diet pills
For Dietrich, the importance of these studies is to show that a single molecule is able to generate very different effects depending on the cell in which it operates. Even in groups of neighboring cells in the same region of the brain, as in the case of AgRP and POMC neurons, mitofusin 2 operates quite differently: AgRP neurons contribute to mitochondrial fusion, while POMC neurons aid in the adhesion of mitochondria to the endoplasmic reticulum.
According to Dietrich, a more general result of this observation is that finding a unique compound that acts on the signaling pathways for hunger and satiety in order to treat obesity will not be simple. Based on data from the Brazilian Institute of Geography and Statistics (IBGE), obesity is an epidemic that currently affects 17% of Brazilians over age 20. In a country with plenty of fatty foods and where unhealthy meals at fast-food restaurants are a common solution to everyday life on-the-go, the findings from Dietrich and his colleagues take on an urgent tone. “The hunger reflex is one of the most basic to survival, one that can not be overcome without putting one’s own life at risk,” says Dietrich.
That’s why, in his view, it is so difficult to develop obesity drugs that do not have serious side effects, as he and Horvath indicated in a 2012 review article published in Nature Reviews Drug Discovery.
But this difficulty has not prevented the search from continuing. On the contrary, it has been stimulated. At the Federal University of São Paulo (Unifesp), a group led by molecular biologist João Bosco Pesquero has just signed a cooperation agreement with their international colleagues at the German-based Max Delbrück Center for Molecular Medicine (see Pesquisa FAPESP Issue No. 211). Its goal is to accelerate the search for a safe and effective drug against obesity that acts on the satiety neurons.
In an article published in July 2013 in Biological Chemistry, Pesquero’s group tested mice that do not produce kinin B1 receptors, involved in leptin action. In these animals, the researchers found an increased activity in the satiety neurons. “These mice have a different metabolism and are protected from obesity even while consuming a high-fat diet,” says Vicencia Sales, a doctoral student in the Pesquero group and co-author of the article.
The Unifesp and German groups, in partnership with their colleagues in Toulouse, France, are betting on the progress of a compound in the experimental testing phase that blocks kinin B1 receptors. Thus, they hope to increase the sensitivity of the mice to leptin and satiate hunger. But to become a viable medication, in addition to other changes, it would still have to be more stable and be able to cross the blood-brain barrier to reach the brain. This will only be worthwhile, however, if the drug is proven to be safe and effective. The drug does not appear to be harmful to mice in tests performed thus far before testing can begin on humans. “Of course we are always worried about how other neurons might respond to this compound,” says Sales, who wants to devote the rest of her PhD to understanding what the drug does in the body as a whole.
For her, Dietrich’s findings prove the importance of looking more carefully at what happens to the mitochondria in their research model. “It is a very difficult job,” she says. After all, we need to isolate the neurons of a brain as small as that of a mouse, about 2 centimeters, to figure out the electrical activity and ATP production. “The hypothalamus of these mice is roughly 0.3 mm and slightly larger than a grain of sand,” she says. Isolating these cells requires a technique that the Yale group has mastered and that made the difference in Dietrich’s work. The web of neurons involving complex and fundamental mechanisms such as the need for food can certainly only be unraveled by the sum of knowledge, expertise and ideas of many groups. Preferably by working together.
Scientific articles
DIETRICH, M. O. et al. Mitochondrial dynamics controlled by mitofusins regulate AgRP neuronal activity and diet-induced obesity. Cell, v. 155, No. 1, p. 188-99. September 26, 2013.
SCHNEEBERGER, M. et al. Mitofusin 2 in POMC neurons connects ER stress with leptin resistance and energy imbalance. Cell, v. 155, No. 1, p. 172-87. September 26, 2013.
TORRES, H. A. M. et al. Kinin B1 receptor gene ablation affects hypothalamic CART production. Biological Chemistry, v. 394, No. 7, p. 901-8, July 2013.