
Phelcom
Phelcom retinal camera attached to a smartphone; app simulates exam of back of the eye. Background: image of retina produced by the cameraPhelcomThere are 45 million blind people in the world, 1.2 million of whom live in Brazil. Around 75% of these cases could have been avoided or treated if everyone received proper diagnosis and eye care. The challenge could be met, to some extent, by using portable devices connected to smartphones, which would make it easier for poorer, underserved populations to receive eye exams. Two such devices have been designed at startups that were launched by Brazilian researchers fresh out of college. One was developed in São Carlos, São Paulo, and the other in Boston.
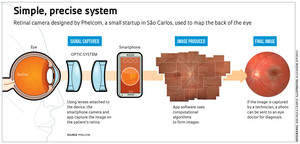
Phelcom was founded by three alumni from the São Carlos campus of the University of São Paulo (USP). For two years, the startup has been working on the handheld Smart Retinal Camera (SRC), a retinal scanner controlled by an attached smartphone. The SRC takes high-resolution images of the back of the retina, or fundus. “The equipment consists of an electronic optical system that connects to a cell phone and an app that controls it all,” explains computer engineer José Augusto Stuchi, one of the project’s creators and Phelcom co-founder. Fundus cameras are a type of ophthalmological equipment that captures images of the back of the eye to diagnose and monitor retinal diseases.

EyeNetra
People line up to use EyeNetra handheld devices in Peru…EyeNetraThe SRC is designed to perform three kinds of fundus exams: color, red-free, and fluorescein angiography (FA). The first two tests check the fundus by capturing high-resolution images of changes to the retina or optic nerve, which are of fundamental importance in the medical monitoring of lesions and degenerative illnesses. FA is performed following the intravenous injection of fluorescein contrast, usually through a vein in the arm or back of the hand. “This is a very fluorescent, non-toxic molecule,” says Diego Lencione, another founding partner of Phelcom, along with Stuchi and Flavio Vieira. “This makes it possible to study the blood flow in the retinal veins and choroid [layer between the retina and sclera], record detailed information on local circulation, and assess functional integrity.”
These three types of exams enable the diagnosis and monitoring of diseases like diabetic retinopathy, glaucoma, age-related macular degeneration (AMD), and retinoblastoma, as well as other eye problems, such as detached retina, edema, and macular hole. The exams generally require traditional tabletop fundus cameras, which are unwieldy and expensive. “These machines are only economically feasible on a large scale, at specialized eye clinics, found just in big cities,” says Stuchi. “That’s why our SRC offers some advantages as a replacement.” Among these advantages, he lists access to eye care in towns that have no clinic, which cuts travel costs for patients.

EyeNetra
…eye exams in AfricaEyeNetraStuchi also says that the SRC makes it easier to test children, people who are bedridden or have limited mobility, and the elderly. “One of the distinguishing features of our device is its ability to capture high-resolution panoramic images of the retina and thus facilitate diagnosis,” adds Stuchi. “Medical reports, evaluations, and case studies can also be performed remotely, eliminating the need for a specialist to travel, since the exam can be done by a technician.” Among disadvantages, Stuchi observes that the SRC is relatively new to the medical market, which may discourage potential buyers. The device will also face indirect competition from more traditional, pioneer brands on the ophthalmological market.
According to Stuchi, the SRC project was inspired in part by the personal experience of one of the partners, Lencione, who has a brother with vision problems related to a retinal disorder. In moving forward with the idea, the team first conducted a market survey, identified a business opportunity, and then initiated technical development. The SRC proof of concept was completed in late 2015. “This enabled us to request funding from PIPE (Innovative Research in Small Businesses Program),” says Stuchi. “As a result, our idea turned into something much bigger. In April 2016, we quit our jobs and founded Phelcom. We are now working on a minimum viable product (MVP) for the SRC so we can begin the first clinical tests.”

EyeNetra
Device for measuring eyeglass prescription…EyeNetraThe product is expected to reach the market in 2018, after it is certified by the Brazilian Health Surveillance Agency (Anvisa). The price tag on the system may be about 10 times lower than that of a standard fundus camera, which is larger, hard to transport, and can perform other functions. Depending on the brand and model, this equipment can cost upwards of R$100,000.
In the opinion of Paulo Schor, professor in the Department of Ophthalmology, of the Federal University of São Paulo (Unifesp), devices that rely on smartphones to perform eye exams do not belong to the future but to the present. “They’re accessible – that is, easy to operate and cheap,” he argues. “This makes them part of a worldwide trend in technology that you just can’t argue with.” Still, he says that society and even ophthalmologists display tremendous innate resistance to the equipment. “This always happens with anything new – with anything that changes what we’re used to,” he explains. “Today it’s standard procedure for patients to have their exams done at an office or clinic. But the trend going forward will be for them to do their own exams and take the results to be evaluated by a doctor.”

EyeNetra
… and device for testing degree of nearsightedness, stigmatism, and farsightednessEyeNetraSchor points out that smartphone devices do not perform quite like traditional fundus cameras. “The tabletop version has specific features, like filters for fluorescein angiography, which is an exam that detects minor leaks in arteries and veins or intraretinal edema,” he says. “Other equipment that is not available in mobile form can also be coupled to standard devices, like optical coherence tomography (OCT) scanners, which do in-depth evaluations of retinal structures, including the choroid.” Nevertheless, Schor does not discard the idea of smartphones. “It depends on what we’re after,” he says. “If their performance is 50% poorer but they’re 400% cheaper and reach 1,000% more people, then they’re a better option. They can screen many more patients than standard equipment and flag for more detailed testing only those who really need it.”
One indication that the SRC is a promising device is the fact that the project took first place in September 2016 at the Brazilian edition of the Falling Walls Lab, an interdisciplinary forum sponsored by the not-for-profit Falling Walls Foundation, based in Germany, which rewards innovative projects by students, researchers, and other professionals.
 Virtual reality
Virtual reality
Work has progressed even further at EyeNetra, founded in Boston in 2011 by Vitor Pamplona, a computer scientist from Santa Catarina. Pamplona developed a kit that contains four products, two of which attach to smartphones (see Pesquisa FAPESP Issue nº 184). This kit has a different purpose than the Phelcom SRC. “The first product performs an eye test for prescribing glasses,” he says. “It’s a game that is installed on the smartphone and uses virtual reality to measure the degree of nearsightedness, astigmatism, and farsightedness and prescribe proper glasses. The game takes two to three minutes to play. It ends with the prescription, which has to be evaluated by an eye doctor.”
The second product is intended for people who already wear glasses and want to know whether they are using the right prescription. The third device is equipped with a set of actual lenses that check the results of the first tests. “With these three products, you can verify what your vision would be like with the prescribed corrections,” explains Pamplona. The fourth product is a set of software programs that collect and store information from the other three and make them available for doctors. So far, EyeNetra has sold some 2,700 kits in 57 countries at a price of $3,000 each. According to Pamplona, he founded the company in partnership with Ramesh Raskar, from India, who was then his professor at the Massachusetts Institute of Technology (MIT), and David Schafran, an MIT researcher.
In England, Andrew Bastawrous, researcher and ophthalmologist at the London School of Hygiene and Tropical Medicine, developed the Portable Eye Examination Kit, called Peek Retina for short. Serving the same purpose as the Phelcom SRC, Peek Retina uses a smartphone camera to perform eye exams that can detect cataracts and retinal diseases. The device can do the exams anywhere and the information is sent to a doctor by email or text message so that a diagnosis can be provided and treatment prescribed. The device is currently undergoing testing in some African nations.
Project
Portable smartphone-controlled equipment for retina diagnosis (nº 2016/00985-5); Grant Mechanism Innovative Research in Small Businesses Program (PIPE); Principal Investigator Flavio Pascoal Vieira (Phelcom); Investment R$225,351.29 and $29,389.58.