 Ana Paula CamposThe brains of four women between the ages of 80 and 82 who died recently in São Paulo provide a few more clues about the complexity of Alzheimer’s disease. Samples from these brains, which were donated to the University of São Paulo (USP) brain bank, when microscopically analyzed revealed the mass of protein plaques and tangles that are typical of the advanced stages of Alzheimer’s. It was thus anticipated that these women during their last decade of life would have suffered from serious problems associated with loss of memory and cognition, such as difficulty in expressing themselves and perceiving the space around them. Interviews with family members and their caregivers, however, found that they remained lucid until the end.
Ana Paula CamposThe brains of four women between the ages of 80 and 82 who died recently in São Paulo provide a few more clues about the complexity of Alzheimer’s disease. Samples from these brains, which were donated to the University of São Paulo (USP) brain bank, when microscopically analyzed revealed the mass of protein plaques and tangles that are typical of the advanced stages of Alzheimer’s. It was thus anticipated that these women during their last decade of life would have suffered from serious problems associated with loss of memory and cognition, such as difficulty in expressing themselves and perceiving the space around them. Interviews with family members and their caregivers, however, found that they remained lucid until the end.
“No one understands exactly why these women did not develop dementia,” admits Carlos Humberto Andrade-Moraes, a neuroanatomist at the Federal University of Rio de Janeiro (UFRJ). His doctorate, under the guidance of Roberto Lent, a neuroscientist at the same university, is the world’s first to analyze the total number of brain cells of the elderly known as asymptomatic Alzheimer’s patients. The study, published with other UFRJ and USP researchers in the December 2013 issue of the journal Brain, found that the number of neurons in asymptomatic patients is practically equal to that of the healthy elderly, unlike what is seen in people with Alzheimer’s who develop dementia, memory loss and loss of cognitive ability. Dementia is characterized by a drastic reduction in the neurons of the hippocampus and cortex, the regions of the brain responsible for memory consolidation and reasoning.
On average, one in 10 people over the age of 65 presents clinical signs of Alzheimer’s. The disease manifests itself first with small memory lapses, which over time become more frequent, followed by failures in moral judgment, space and time perception, and increased difficulty communicating. The average survival rate is eight years, during which time symptoms worsen to total incapacitation.
It has been known for some time that dementia is caused by the destruction of synapses, the trillions of connections among 86 billion neurons, the brain cells that store and transmit information, from which memories and thoughts emerge. A healthy neuron receives up to 10,000 synapses from other neurons, exchanging electrical signals and substances that keep it alive. In Alzheimer’s disease, the neurons atrophy and die, because they are unable to maintain the synapses. Consequently, the volume of the hippocampus and the thickness of the cortex decrease, which can be seen in magnetic resonance images. According to Dr. Marcio Balthazar, a neurologist who treats Alzheimer’s patients at the Hospital das Clínicas of the University of Campinas (Unicamp), neuroimaging may help in diagnosing the disease, but it cannot replace laboratory, clinical and psychological tests.
In partnership with Dr. Fernando Cendes, a neurologist at Unicamp, Dr. Balthazar and his collaborators are hoping to perfect a new way to identify Alzheimer’s at an earlier stage: the use of neuroimaging to assess brain activity, not just the brain’s anatomy. The idea is to observe functional brain activity in magnetic resonance images when patients are relaxed and not thinking about anything. “Even when the patient is at rest, we see that some areas of the brain are simultaneously activated, pulsing at the same frequency, which suggests that groups of neurons are communicating,” explains Dr. Balthazar. “In a patient with Alzheimer’s, this network is less connected.”
 In an article published in the November 2013 issue of Psychiatric Research: Neuroimaging, the Unicamp group was able to distinguish the neuroimages of brain activity at rest in people with mild dementia symptoms from those of healthy elderly, with about 70% accuracy. The researchers also observed a relationship between the number of network connection failures and the degree of memory loss.
In an article published in the November 2013 issue of Psychiatric Research: Neuroimaging, the Unicamp group was able to distinguish the neuroimages of brain activity at rest in people with mild dementia symptoms from those of healthy elderly, with about 70% accuracy. The researchers also observed a relationship between the number of network connection failures and the degree of memory loss.
The sooner the better
“We hope to improve the method for making a diagnosis much earlier,” Dr. Balthazar says. Although Alzheimer’s remains incurable, the sooner the diagnosis is made the more effective are the interventions to alleviate the symptoms: the use of acetylcholinesterase inhibitors, occupational therapy, psychological rehabilitation and physical activity, in addition to family planning for the future.
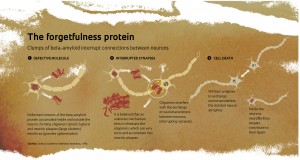
Since senile dementia may have other causes—vascular problems and other degenerative diseases—an Alzheimer’s diagnosis is usually confirmed only after death. The autopsy of brain tissue reveals an excess of so-called neuritic plaques, anchored in branches of neurons, and neurofibrillary tangles within the atrophied neurons. These signs are found especially in the hippocampus and cerebral cortex. Until a few years ago, most researchers believed that the neuritic plaques were responsible for synaptic dysfunction. But recent studies done by the team of Fernanda De Felice and Sergio Teixeira Ferreira, respectively a UFRJ neuroscientist and biochemist, are demonstrating that the plaques, while toxic, are not the major cause of synaptic elimination and the death of neurons (see Pesquisa FAPESP Issue nº 157).
In fact, the plaques are formed by the accumulation of small molecules of beta-amyloid. Normally produced by the brain, this protein undergoes deformation in Alzheimer’s. But today many researchers believe there are much smaller clumps of beta-amyloid—oligomers, which are able to circulate inside and outside neurons, and are responsible for synaptic interference. Other studies suggests that these oligomers also form neurofibrillary tangles, which prevent the transport of substances within the neurons and contribute to their death. According to this reasoning, the formation of plaques is the body’s attempt to sweep the oligomers outside the cells and away from the synapses. “The plaques would be protective and not the cause of dementia,” says Andrade-Moraes.
The discovery of asymptomatic Alzheimer’s patients reinforced this hypothesis. These cases appeared and were first described in studies that followed hundreds of elderly people in the United States. A comparison of the clinical tests to which these people were regularly subjected, along with the postmortem analysis of their brains, showed that 25% to 40% of the cases histologically diagnosed as having Alzheimer’s had not developed dementia. “Although it is doubtful whether these individuals would have remained clinically normal if they had lived longer, they seem to have been able to offset or delay the onset of symptoms of dementia,” wrote Dr. Juan Troncoso in 2012, a neuropathologist at Johns Hopkins University and one of the first to draw attention to asymptomatic patients.
According to Andrade-Moraes, before the study that was recently published in Brain, no work on the impact Alzheimer’s has on the number of brain cells made a comparison between individuals with and without dementia. “We wanted to know if asymptomatic people had any change in the composition of their brain cells,” he says.
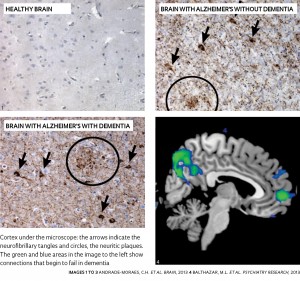 ANDRADE MORAES, C.H. ET AL. ET AL. BRAIN 2013 4: BALTHAZAR M.L. ET AL. ET AL. PSYCHIATRY RESEARCH 2013 The study was conducted in partnership with the team led by Dr. Lea Grinberg, a neuropathologist and coordinator of the USP Human Brain Bank, which, besides analyzing the brains of the elderly who died in São Paulo, investigates, through family and caregiver questionnaires, the cognitive performance of these people up to 10 years prior to death.
ANDRADE MORAES, C.H. ET AL. ET AL. BRAIN 2013 4: BALTHAZAR M.L. ET AL. ET AL. PSYCHIATRY RESEARCH 2013 The study was conducted in partnership with the team led by Dr. Lea Grinberg, a neuropathologist and coordinator of the USP Human Brain Bank, which, besides analyzing the brains of the elderly who died in São Paulo, investigates, through family and caregiver questionnaires, the cognitive performance of these people up to 10 years prior to death.
The USP and UFRJ researchers selected 14 brains from women who died between the ages of 71 and 88 (Alzheimer’s prevalence is slightly higher among women). Five had a level of plaques considered normal for their age, while others presented the characteristic excess of Alzheimer’s. Of the latter, five had signs of dementia and four were asymptomatic.
Fewer neurons, more glia
The brains were processed at UFRJ in an automatic isotropic fractionator machine, built by Lent’s team (see Pesquisa FAPESP Issue nº 192). The machine transforms portions of the brain into a homogeneous suspension containing cell nuclei. Colored antibodies that bind to the nucleus of neurons allow them to be distinguished from other brain cells, the glial cells.
As expected, the hippocampi of the women with dementia had half the number of neurons found in the hippocampi of the healthy asymptomatic women; those with dementia also had fewer neurons throughout the cortex. At the same time, the brains of people with dementia had a higher proportion of glial cells (see the infographic on page 50). “These cells increase in number to protect neurons, but as the disease progresses, they cause an inflammation that worsens the symptoms of dementia,” Andrade-Moraes explains. He, however, found no significant difference in the number of neurons and glial cells between the brains of the healthy elderly and the elderly with asymptomatic Alzheimer’s.
“The asymptomatics must have some unknown physiological mechanism that protects their neuritic network from the effects of the oligomers,” he suspects. “Something separates the oligomers from the synapses, as they rapidly accumulate into plaques.”
Andrade-Moraes thinks there is one candidate to explain this mechanism: the more efficient action of insulin in the asymptomatic brain. Unlike what occurs in other organs, the role of insulin in the brain does not appear to be to control sugar metabolism, but rather memory consolidation and the formation of new synapses. In-vitro experiments and animal experiments conducted by Fernanda De Felice and Sérgio Ferreira have shown that insulin protects neurons from oligomeric action. In an article published in the December 2013 issue of Cell Metabolism, they presented new neuronal mechanisms that cause synaptic loss in mice and monkeys with signs similar to those of Alzheimer’s. As part of his doctoral work, Mychael Lourenço showed that a drug used to treat type 2 diabetes, liraglutide, blocked neuronal damage in animal models of Alzheimer’s. Currently a team at London’s Imperial College is testing liraglutide on 200 people with Alzheimer’s.
Another hypothesis is that those who are asymptomatic have greater cognitive reserves, perhaps the result of a synaptic network that is more complex than that of those who develop dementia. These reserves would put up more resistance to the effects of the oligomers. This idea comes from the observation that asymptomatic people usually have a higher level of education, or learned to speak and write in early childhood. At Unicamp, Dr. Balthazar is trying to confirm the protective effect of cognitive reserves, by comparing the connectivity of neural networks in elderly patients with varying degrees of education, reading habits and social life.
Projects
Brazilian Institute of Neuroscience and Neurotechnology – BRAINN (nº 2013/07559-3); Grant Mechanism Research, Innovation and Dissemination Centers (RIDC); Coord. Fernando Cendes – FCM/Unicamp; Investment R$ 13,621,302.32 (FAPESP).
Scientific articles
ANDRADE-MORAES, C.H. et al. Cell number changes in Alzheimer’s disease relate to dementia, not to plaques and tangles. Brain. December 2013.
LOURENCO, M.V. et al. TNF-alpha Mediates PKR-dependent memory impairment and brain IRS-1 inhibition induced by Alzheimer’s beta-amyloid oligomers in mice and monkeys. Cell Metabolism. December 3, 2013.
BALTHAZAR, M.L. et al. Whole cortical and default mode network functional connectivity mean the potential biomarkers for mild Alzheimer’s disease. Psychiatry Research. November 11, 2013.