Rodrigo Méxas and Raquel Portugal / Fiocruz
Female mosquito Aedes aegypti, the main transmitter of Zika and three other viruses in BrazilRodrigo Méxas and Raquel Portugal / FiocruzIn late 2016, the idea began to take hold that microcephaly represents only a fraction of the problems caused by the Zika virus—possibly the smallest, but the most tragic because of its long-term consequences. Papers published in December 2016, and the months before that, began to characterize in much more detail the broad spectrum of damage that the infected infant can undergo in pregnancy: from the most serious and incompatible with life to unperceived subtle damage that perhaps could lead to a full life. In addition to these extremes, which are rare, a broad range of lesions may appear in the central nervous system—the most common are calcifications, a kind of brain scar tissue—capable of compromising the development and independence of the infant to an uncertain degree. According to experts, this continuum of effects reinforces the idea put forth some time ago that the virus causes a syndrome: congenital Zika syndrome.
From the time the virus was suspected of causing the cases of microcephaly to characterization of the syndrome attributed to it, a relatively short period of time had transpired in terms of scientific research, an activity that tends to work at a slower pace, marked by the availability of money and infrastructure for the experiments. The alarm was sounded in Recife’s maternity wards in early August 2015 when pediatric neurologist Vanessa van Der Linden and other doctors in the northeastern state of Pernambuco began to identify an atypical rise in microcephaly cases. The following month, the Ministry of Health asked epidemiologist Celina Turchi Martelli of the Aggeu Magalhães Research Center in Recife to investigate the problem. She contacted researchers in Brazil and abroad and created a task force to study the connection between increased microcephaly and the Zika infection—for her work, Martelli was selected by the journal Nature last December as one of the 10 researchers who mattered most in 2016.
Three months after seeing an increase in microcephaly cases, evidence that the Zika virus was causing the problem became stronger. In mid-November 2015, obstetrician Adriana Melo, with the help of researchers from the Oswaldo Cruz Foundation (Fiocruz) in Rio de Janeiro, identified the virus in the amniotic fluid of two pregnant women from Paraíba (northeast Brazil), whose fetuses had microcephaly. In late November 2015, the team led by virologist Pedro Vasconcelos of the Evandro Chagas Institute (IEC) in Pará isolated the virus from several tissues of a baby from Ceará (northeast Brazil) who was born with microcephaly and died shortly after birth.
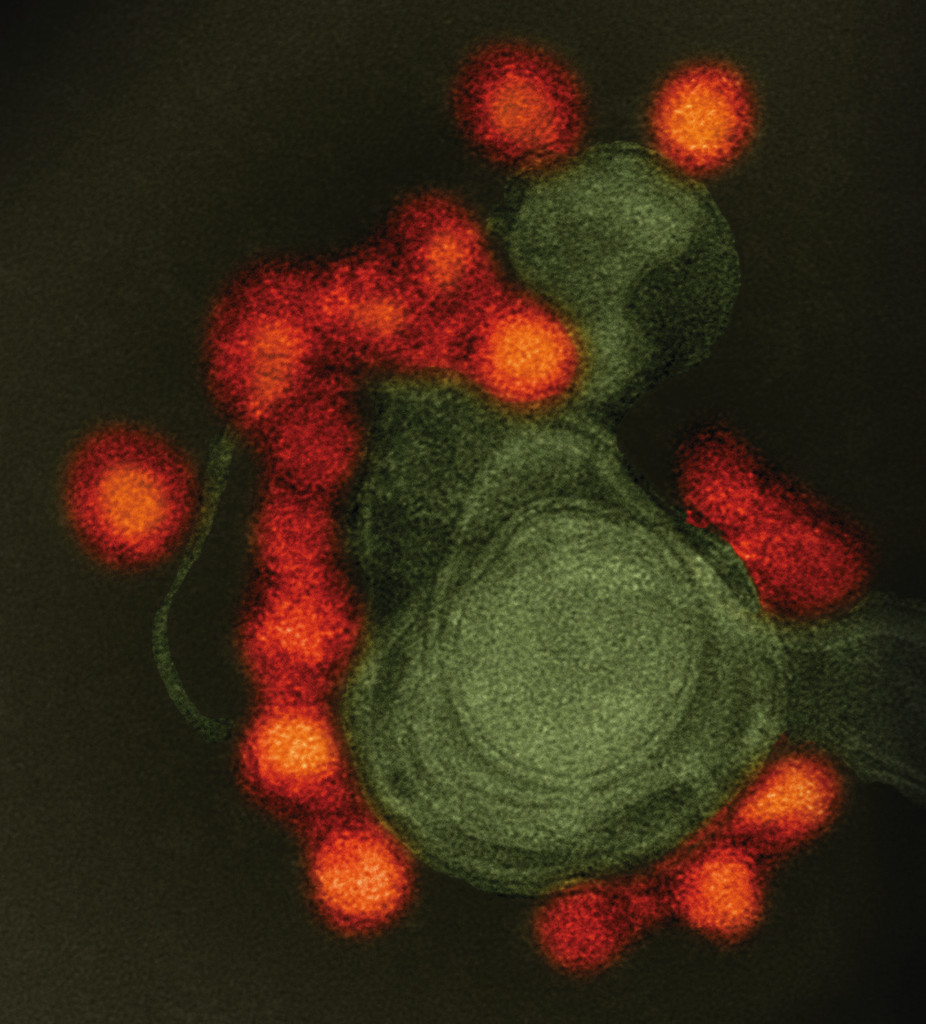
National Institute of Allergy and Infectious Diseases, National Institutes of Health
Isolated Zika copies (in red) of an infant born with microcephaly in FortalezaNational Institute of Allergy and Infectious Diseases, National Institutes of Health“After the initial tests in which we identified the viruses, I had no doubt that Zika was causing microcephaly,” says Pedro Vasconcelos, director of the IEC, where he heads up a laboratory that serves as a national reference center for arboviruses, diseases caused by viruses transmitted by arthropods, such as Zika, dengue and yellow fever. “I was convinced that we had done good work,” Vasconcelos said in mid-May 2016 during a trip to Rio de Janeiro. “I was at a meeting in Brasilia in late October 2015 and there was a lot of chatter. I said that if I were sent enough of the right material, in two or three days I would have a diagnosis,” he says. And so he did.
In mid-November 2015, pathologist Fernanda Montenegro de Carvalho Araújo, from the Central Laboratory of Public Health (Lacen) in Ceará, sent Vasconcelos tissue samples from a baby with microcephaly. A few days later, Vasconcelos informed the Ministry of Health that, on November 28, he had found the connection between the Zika infection in pregnancy and cases of microcephaly. A press release from the Ministry reported that it was “an unprecedented situation for world scientific research” and called for a national mobilization to contain Aedes aegypti, the main transmitter of the Zika and dengue viruses and the newcomer, chikungunya fever, which, at that time, had researchers and health officials more worried.
Having isolated the virus, Vasconcelos distributed samples to teams in Brazil and abroad in order to speed up the research. The next few months were very hectic. They needed to learn how to maintain and multiply the virus and generate enough copies for the experiments. At the Institute of Biomedical Sciences (ICB) of the University of São Paulo (USP), virology groups led by Edison Durigon and Paolo Zanotto immediately attempted to replicate the virus and, in partnership with the group led by neuroimmunologist Jean Pierre Peron, also of ICB, and neuroscientist Patrícia Beltrão-Braga of the USP School of Veterinary Medicine and Animal Husbandry (FMVZ), began experiments using mice and stem cells grown in a three-dimensional matrix that allows different cell types to be generated and organized into microscopic spheres (neurospheres) or layered like mini-brains.
Léo Ramos Chaves
Cell culture used to produce copies of the virus in the Edison Durigon laboratory at USPLéo Ramos ChavesThe aim of these experiments was to better understand how the virus was transmitted from the mother to the fetus, how it behaved within the fetus, and during what period of the pregnancy were women most vulnerable. They also needed to confirm that it was the Zika virus causing the problems, which is what researchers call a causal relationship, because they had not yet ruled out the possibility that the virus could be in the brain without causing damage.
Much evidence began to accumulate in the following months. In February 2016, Slovenian researchers found the virus and quantified its presence in the brain of an 8-month-old fetus, aborted by a woman who had been pregnant while living in Rio Grande do Norte State and who had shown signs of Zika during her pregnancy. This discovery was followed by others that showed the virus was capable of crossing the placenta and infecting the amniotic fluid, something few viruses can do.
With the attention of research groups in Brazil and abroad focused on Zika, new developments emerged almost daily. In Brazil, advances made in laboratories were published before the results had been analyzed and judged by other scientists, reversing the usual flow of information characteristic of scientific production. Following a guideline announced in September 2015 by the World Health Organization (WHO) and agreed to by some scientific journal publishers, many researchers made the work of their groups available as quickly as possible.

Up until 2015, there were two articles on Zika written by Brazilians: the first analyzed the genetic changes of the virus…
Two studies presented in early March 2016 demonstrated that Zika was capable of infecting and killing human neural cells. In the first study, researchers from Rio de Janeiro and São Paulo, working under the coordination of Stevens Rehen and Patricia Garcez, neuroscientists at the D’Or Institute for Research and Education (IDOR) and the Federal University of Rio de Janeiro (UFRJ), found that the virus preferred to invade and kill neural progenitors, the precursor stem cells of neurons, and glial cells, which make up nervous tissue. A few days later, researchers from the United States confirmed this finding and showed that Zika was infecting in vitro neural cells similar to those that make up the cortex, the outermost layer of the brain, associated with cognitive functions such as attention, memory and language.
Almost at the same time, the USP, ICB and FMVZ groups achieved similar results in neurosphere and mini-brain experiments. In the same study, carried out with the participation of researchers from the Pasteur Institute in Senegal and published May 11, 2016 in Nature, the São Paulo researchers also presented the first animal model of microcephaly caused by Zika and established the causal relationship between the virus and microcephaly.
Using copies of the virus produced in the Durigon and Zanotto laboratories, Peron and his group inoculated pregnant mice with the Zika variety in circulation in Brazil. The virus crossed the placenta of the females of a strain more susceptible to viral infections and impaired the development of the pups. They had lower birth weights, smaller brains and damage of the kind seen in human infants (see Pesquisa FAPESP Issues nos 242 and 244). This microcephaly model was then used in tests conducted by researchers from the United States, in partnership with USP, who demonstrated the potential for developing a vaccine against Zika.
A warning sign
In view of these results and the increase in suspected cases of microcephaly in Brazil, on March 31, 2016, WHO issued a report in which it asserted that there was a “strong scientific consensus” that the Zika virus caused microcephaly and Guillain-Barré syndrome, a disorder that leads to the destruction of myelin, a substance forming the sheath that lines the nerves, and can cause paralysis. It was a major change of the position expressed just two months before, when WHO had said there was a “possible association” of the virus with these problems. It now declared an international public health emergency.
“Since the beginning of the epidemic, knowledge of the virus has grown considerably, especially about its biology and interaction with mammalian organisms,” says Peron. The year 2016 produced more information about the Zika virus than had come to light in the previous six decades. Pubmed, one of the most important databases indexing scientific articles in biology and life sciences, alone recorded 1,756 articles on Zika in 2016 (almost 200 of which were written by Brazilians). This total is approximately eight times larger than the volume available by the end of 2015. Beginning in 1952, when the first human infection was recorded, up until the virus became a worldwide concern in 2015, there were 218 articles on Zika in Pubmed. Some studies showed Zika’s affinity for the mammalian nervous system, and several others described sporadic cases of fever in Africa and Asia. Despite the 2007 epidemic in the Yap Islands in Micronesia and the 2013 epidemic in French Polynesia, when thousands were infected, nothing was known about Zika’s action on developing fetuses.
“The outbreak in Micronesia should have served as a warning,” says Zanotto. “I’m engaging in this self-criticism because, at the time, we, arbovirus specialists, were sequencing African varieties of Zika, but we never imagined that the virus could change its performance profile so quickly and drastically.” He says that, at the time, he and other experts were eyeing other arboviruses considered to be of greater risk and pandemic potential, such as Rift Valley fever, strain 2 of the West Nile fever virus, and the Chikungunya fever virus. “Today there is a consensus in the research community that we need to take a more proactive approach to emerging pathogens,” says Zanotto, who, in late 2015, began to mobilize the Zika Network, a consortium of São Paulo researchers who are investigating the virus with the support of FAPESP.

Pan American Health Organization PAHO
Participant in a study that followed pregnant women with symptoms of Zika in RecifePan American Health Organization PAHOIn addition to collaborating on development of the microcephaly animal model, he is working with Durigon and biologist Luis Carlos de Souza Ferreira, also of USP, to improve a serological test capable of distinguishing a dengue infection from one caused by Zika. Zanotto co-authored one of the two papers by Brazilian researchers among the 218 articles about Zika that were published before 2015. In that study, published in January 2014 in PLOS Neglected Tropical Diseases, Zanotto’s team and the group led by Senegalese virologist Amadou Alpha Sall of the Pasteur Institute in Dakar, sequenced the genetic material of Zika samples collected in Africa and Asia to study the changes that the virus had undergone since it was isolated in 1947. The work later made it possible to reconstruct the migratory route of the virus (via Asia) to Brazil and verified that along the way it seems to have adapted to infect humans.
One virus, multiple kinds of damage
While some of the researchers were engaged in learning more about the biology of the virus, other groups concentrated on characterizing the congenital syndrome caused by Zika. In January 2016, a group coordinated by Dr. Lavinia Schüler-Faccini, a geneticist at the Federal University of Rio Grande do Sul (UFRGS), conducted a series of tests on some of the first children born with Zika-related microcephaly from eight Brazilian states and found that microcephaly was not the only problem. Many presented other neurological changes, such as calcifications; lissencephaly, which is characterized by the absence of the usual folds of a healthy brain; and pachyderm, characterized by more extensive folds; as well as a severe joint problem called arthrogryposis.

Léo Ramos Chaves
Mini-brains grown at USP and used in Zika experimentsLéo Ramos ChavesIn late August 2016, an article published online in the journal Radiology and authored by researchers from São Paulo, Paraíba, Rio de Janeiro and Campinas, provided the results of the evaluation of 17 infants who were infected with Zika during pregnancy and 28 others suspected of having had contact with the virus. All of the infants presented reduced cerebral volume, which was more pronounced among those with confirmed infection, according to the study, coordinated by radiologist Dr. Fernanda Tovar-Moll, a professor at UFRJ and a researcher at IDOR. In some cases, the researchers found brains that had, in fact, developed normally in early gestation but, over time, grew more slowly.
Two months later, pediatric neurologists André Pessoa, from Ceará State, and Vanessa van der Linden, from Pernambuco State, reported 13 cases of infants infected by Zika during pregnancy and born with borderline cephalic circumference and neurological changes detected by imaging exams. A few months after childbirth, 11 presented skulls smaller than normal for that age—they had developed postnatal microcephaly—because of a slowing of brain development. “The absence of microcephaly at birth does not exclude congenital Zika infection nor the presence of abnormalities in the brain or other organs related to Zika,” the researchers wrote in an article in Morbidity and Mortality Weekly Report.
In the December 15, 2016 edition of the New England Journal of Medicine, infectious disease specialist Patrícia Brasil and her team at Fiocruz in Rio de Janeiro, in partnership with collaborators in São Paulo, the United States, Sweden and Austria, described the outcome of what is probably the most comprehensive study following pregnant women with Zika in Brazil. In the study, they presented the results of the pregnancies of 186 women (125 positive for Zika and 61 negative) of the 345 who have been followed in the city of Rio de Janeiro since the height of the Zika epidemic in Brazil.
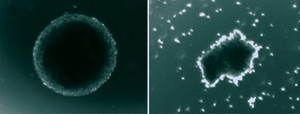
Pan Patricia Garcez / UFRJ and IDOR
Neurosphere formed from healthy cells (left) and another produced by virus infected cellsPan Patricia Garcez / UFRJ and IDOROf the 117 infants exposed to Zika during pregnancy and evaluated by researchers, only four (3.4%) were born with microcephaly and 49, equivalent to 42% of the total, showed signs of abnormal development in the first month of life, almost always associated with damage to the central nervous system. Imaging studies showed calcifications in the nervous system of several of the 49 infants and reduction in the size of the brain—but not necessarily of the skull—as well as an increase in the volume of brain cavities (ventricles). These changes occurred in infants whose mothers were infected at the beginning, middle and end of their pregnancies—one was infected by Zika in the 39th week, shortly before delivery. These observations indicate that the virus poses a risk to the fetus throughout the pregnancy.
Taken together, these results help explain why women infected in pregnancy do not always give birth to babies with microcephaly. In São José do Rio Preto, in inland São Paulo State, the team led by virologist Dr. Maurício Nogueira began to follow 1,200 pregnant women in early 2016. Of these, only 54 had the Zika infection. None of the babies, however, were born with microcephaly, although 30% had neurological lesions. In Jundiaí, a city 60 km from São Paulo, the pediatric group led by Saulo Duarte Passos has been following 560 pregnant women since March 2016. As of December 2016, 265 babies had been born and 33 had microcephaly—just three cases were proven to be caused by Zika, the others are being analyzed. Among those born with normal heads, some presented neurological, visual and auditory changes around the 5th or 6th month after birth.
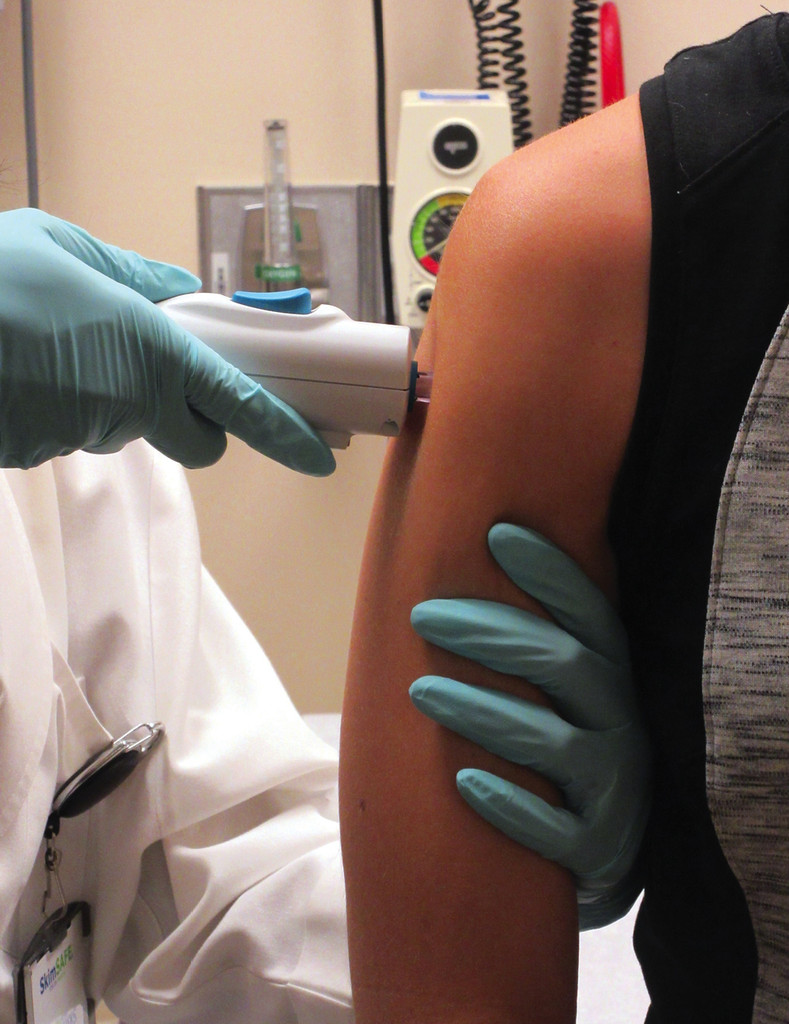
National Institute of Allergy and Infectious Diseases, National Institutes of Health
Volunteer receives anti-Zika vaccine in initial tests with humans conducted in the United StatesNational Institute of Allergy and Infectious Diseases, National Institutes of HealthA search for immunity
At the Center for Virology and Vaccine Research (CVVR) of Harvard Medical School in the United States, the group led by Dan Barouch, in which Brazilian immunologist Rafael Larocca participates, tested two formulations in mice and proved, in partnership with the ICB-USP groups, that they protected the animals from Zika infection. Two months later the test results on monkeys came back, and clinical trials of two vaccine candidate compounds on humans began. Today, there are about 30 formulations in different stages of evaluation. Although they were shown to be effective in humans, it could take years before they are available for use by the general population.
“It was amazing to see how responsive Brazilian science was,” says Rehen, research director at IDOR. He believes this was possible because over a period of almost 10 years, large sums of money were made available for research in Brazil, resulting in an installed capacity to carry out the investigations. “The existence of a real problem forced the scientific community to organize in search of solutions,” he added. “We showed we could do it.”
Republish