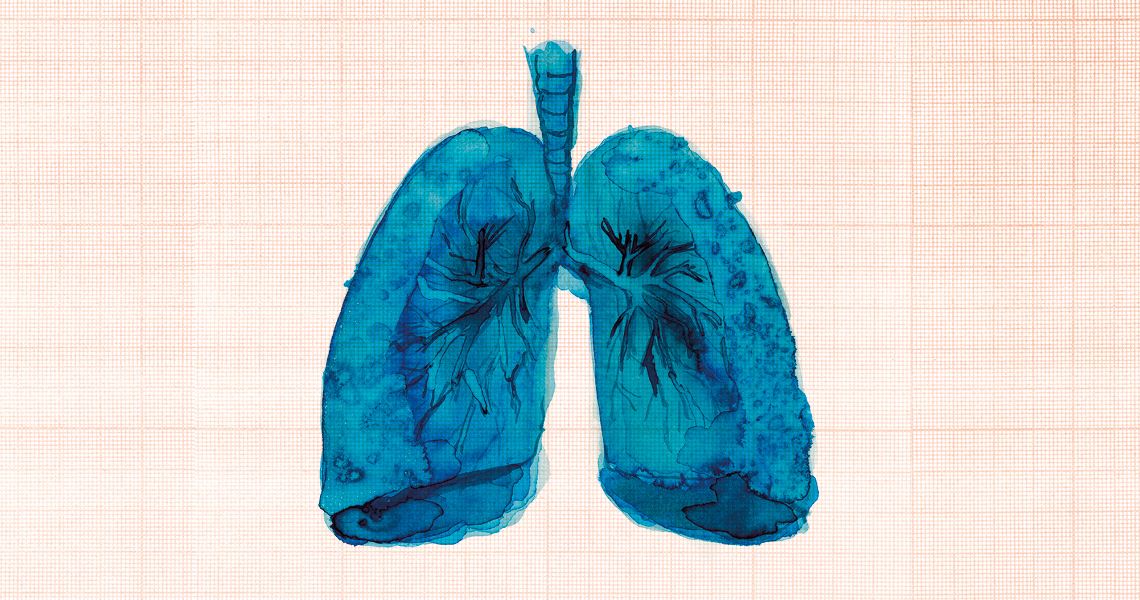
Almost nine months after the pandemic began, our accumulated knowledge of COVID-19, the disease caused by the novel coronavirus (SARS-CoV-2), suggests that its harmful effects on humans may be greater and more lasting than first thought. Previously described as a slightly more severe pneumonia that manifests itself in patients with serious symptoms, COVID-19 is now considered by physicians and researchers to be a more wide-reaching disease, capable of triggering a broad inflammatory response similar to that caused by sepsis. “The lungs are the infection’s ground zero,” says pathologist Marisa Dolhnikoff, leader of a team at the School of Medicine (FM) of the University of São Paulo (USP) which is performing autopsies on people who die from COVID-19. “We know that the virus is capable of infecting cells in other organs, such as the heart, kidneys, and the central nervous system.”
This and the clinical cases examined by researchers around the world have drawn attention to the possibility that for some patients, certain symptoms of COVID-19 may persist long after the acute phase of the disease ends. People with severe and moderate cases of the disease may go on to develop what is being called post–COVID-19 syndrome. The list of long-term symptoms is extensive and varied, including fatigue, rapid heart rate, shortness of breath, joint pain, persistent loss of smell and taste, and difficulty concentrating. “I have patients who recovered months ago but still haven’t regained their taste; others have lost their sense of smell,” comments pulmonologist Margareth Dalcolmo, from the Oswaldo Cruz Foundation (FIOCRUZ) in Rio de Janeiro. The researcher herself recovered from the disease two months ago, and still suffers from peripheral neuropathy, such as numbness in her hands.
– Potential treatment
– The uncertainty of herd immunity
– Scientific philanthropy boost
– The distance between dental research and treatment
Recent studies also suggest that COVID-19 can cause more serious complications and even make other diseases more likely to occur, such as diabetes, when the body does not properly metabolize sugar (glucose) molecules in the blood. This is a worrying scenario for scientists and raises the question of whether everyone who has overcome COVID-19—more than 16 million people worldwide to date, of which 3 million are in Brazil—can really be considered cured simply for having survived the initial infection. “As a result, it is possible that the number of deaths is indirectly much higher than estimated,” says infectious disease specialist Marcus Vinícius Lacerda, from the Dr. Heitor Vieira Dourado Foundation for Tropical Medicine in Manaus. “Many patients who have recovered from COVID-19 and been discharged from hospital may later die of complications related to the infection. These deaths will not be counted in the pandemic figures, even if they are related.”
The statistics available do not yet paint a clear picture of this new chapter of the pandemic. It is also difficult to estimate a patient’s risk of developing long-term symptoms after overcoming the acute phase of COVID-19, or how long these symptoms might last. Scientists do not know what factors contribute to the risk of a person infected by the novel coronavirus developing more serious complications after the acute stage. Most studies following individuals considered cured of the disease are in the early development stages or are still ongoing. It is no coincidence that most evidence of post–COVID-19 syndrome has come from research in the first countries hit by the pandemic between December 2019 and February 2020, such as Italy. A study published in the Journal of the American Medical Association (Jama) in July found that 87.4% of 143 patients admitted to a hospital in Rome with COVID-19 were still exhibiting symptoms two months after being discharged.
The most common problem identified by the researchers was shortness of breath (dyspnea). This complication results from the formation of fibrosis—small scars—in the lung tissue, which makes the organ less flexible, reducing its ability to exchange gases and oxygenate the body. Pulmonary fibrosis is more frequently seen in the most severe cases of COVID-19, caused directly by the virus as well as indirectly as a consequence of an unregulated inflammatory response in the affected area. Fibrosis can also occur as a result of spending a long time in an ICU breathing with the assistance of a mechanical ventilator. “The risk of fibrosis in the lungs varies according to the patient’s age, preexisting conditions, or even individual genetic factors,” explains Dolhnikoff. “What is clear is that a portion of the affected population develop these scars, which have an impact on their quality of life.”
Kika Carvalho
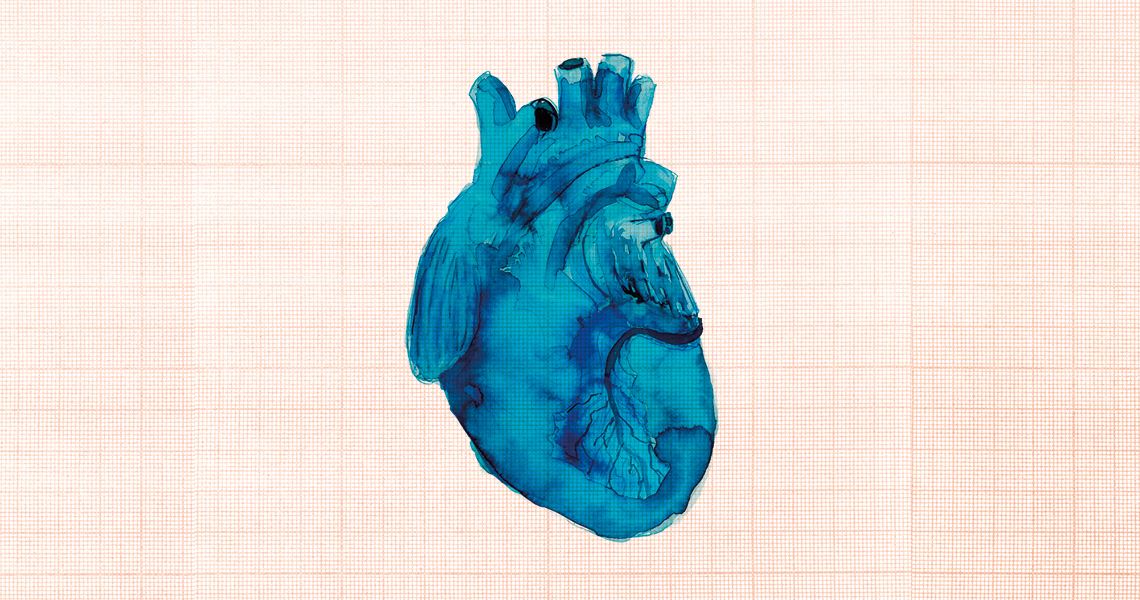
SARS-CoV-2 can also infect the cells that line the inner wall of blood vessels, altering the coagulation mechanism and leading to hypercoagulability, with an increased chance of thrombi that can cause infarctions and hemorrhages in the heartKika CarvalhoLong-term complications can also extend beyond the lungs. In Germany, a study published in the journal Jama Cardiology in July evaluated 100 patients between 45 and 53 years of age. All of them recovered from COVID-19. Ten weeks after diagnosis, however, 78% of them had developed cardiac abnormalities due to inflammation in the heart.
In Brazil, at least two studies are monitoring patients who have survived the disease and the long-term impacts on their health. One is being conducted by Marcus Lacerda and his team, who in July started collecting data from 800 patients discharged from hospitals in Manaus after testing negative for the disease. “We will track those who die after leaving hospital and identify the causes of their deaths,” he explains. The aim is to estimate the incidence of certain health problems in people considered cured, and to compare these numbers with people who have not had COVID-19. “We are also going to monitor those who survive but go on to develop other illnesses or complications, to look for possible relationships between them and COVID-19.”
Kika Carvalho
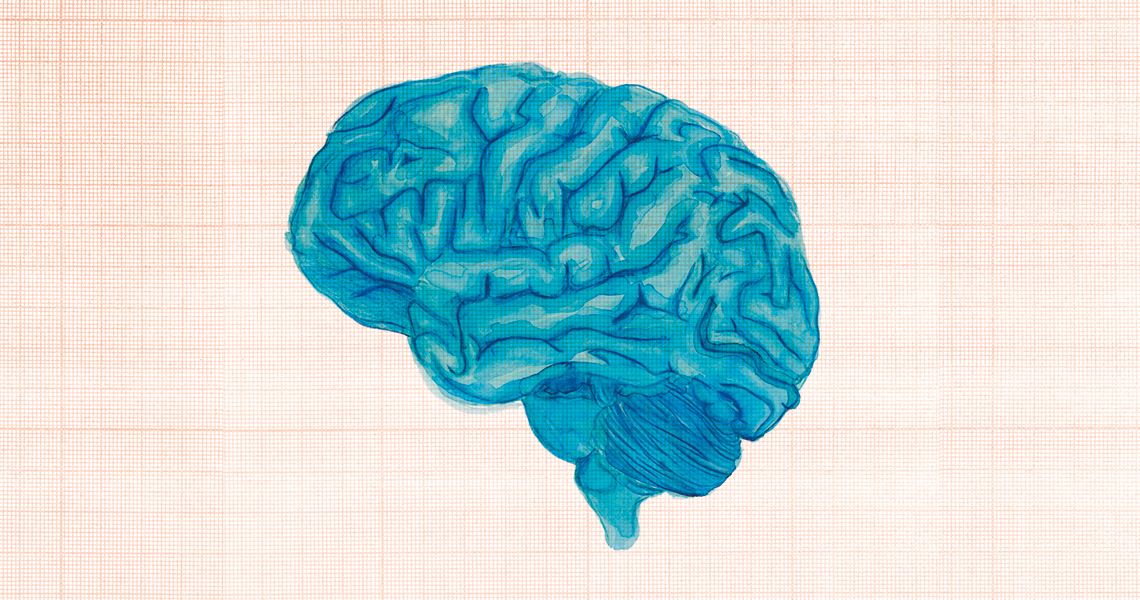
Studies suggest that the novel coronavirus is capable of invading and damaging a range of neuronal cells, such as astrocytes and neurons, causing inflammation that can result in psychomotor agitation, delirium, and hallucinations months after the acute phase of COVID-19 passesKika CarvalhoA team led by physician Augusto César de Oliveira, from the Emílio Ribas Institute of Infection Diseases in São Paulo, also started a study in July, in partnership with the Albert Einstein and Sírio-Libanês hospitals in São Paulo, and Fortaleza General Hospital in Ceara. The objective is to assess the incidence of long-term neurological disorders in individuals infected with SARS-CoV-2. “We will analyze patients over the next six months,” explains the researcher. He says that since the beginning of the pandemic, clinical reports indicate a high incidence of headaches and mental confusion in people who have had COVID-19. “We initially thought this was associated with the acute phase of the disease, but the amount of people still suffering neurological problems after overcoming the disease is surprising.”
Oliveira cites a specific example: a middle-aged woman, with no pre-existing conditions, who was infected with the novel coronavirus and developed a moderate case of COVID-19. “She recovered from the illness, but four months after her first symptoms, she began experiencing cognitive and behavioral changes, such as memory lapses, difficulty concentrating, and feelings of agitation,” says the researcher. SARS-CoV-2 was found in the patient’s cerebrospinal fluid, which surrounds and protects the brain, “suggesting that the virus is not only capable of invading the central nervous system, but of remaining there for a prolonged period of time, even after the primary symptoms of the disease have stopped.” According to physician Jorge Casseb, a researcher at the São Paulo Institute of Tropical Medicine (IMT) at USP and one of the project’s sub-coordinators, “these sequelae can cause potentially serious injuries over a patient’s lifetime.”
Dalcolmo, from FIOCRUZ, has been observing similar cases at his private practice. “I have patients who have overcome the acute phase, but almost a month later developed meningoencephalitis, inflammation that affects the brain and its protective membranes, called the meninges.” Some even tested negative for the virus after providing respiratory secretion samples for a RT-PCR test, considered the gold standard for COVID-19 diagnosis. Yet they later continued to suffer from the symptoms of the disease or developed more serious complications.
The situations observed by Oliveira and Dalcolmo raise an important question: is the virus still present in the bodies of patients with persistent symptoms or more serious complications months after the acute phase of the infection? There is apparently no pattern. Some still carry the virus, even if at lower levels, while others do not. Some experts also suggest that RT-PCR tests cannot detect the virus in other parts of the body, such as the central nervous system, where its effects can lead to the emergence of other diseases. In July, Lacerda and his team described the case of a teenager in Manaus who developed Guillain-Barré syndrome after being infected with the novel coronavirus. This immune disorder damages the myelin, a substance that lines the nerves, causing muscle weakness and in severe cases, total paralysis of the limbs, and even death.
All of these complications demonstrate the virus’s versatility and ability to invade different cell types. Like a key fitting into a lock, SARS-CoV-2 uses its S protein (S for spike) to bind to angiotensin-converting enzyme 2 (ACE2) on the surface of the cells that cover the lung. “It just so happens that the cells of the heart, the central nervous system, and several other organs also express ACE2, making practically all human tissue vulnerable,” explains biomedic Marcelo Mori, from the Institute of Biology (IB) at the University of Campinas (UNICAMP). “After invading the cells, the virus begins to multiply,” says biochemist José Carlos Alves-Filho of the Ribeirão Preto School of Medicine at USP. “After replicating a certain number of times, the copies break through the cell membrane and infect other cells, repeating the process.”
They can even infect monocytes, immune system cells produced in the bone marrow. Monocytes travel via the bloodstream and can differentiate into macrophages, immune cells that detect and engulf foreign particles and microorganisms. In a study published in the journal Cell Metabolism in July, Mori and other researchers from UNICAMP’s COVID-19 task force found that SARS-CoV-2 multiplies faster inside these cells in patients with high blood glucose levels. Glucose is used by cells to produce energy. “In these cases, however, the virus appears to use it to replicate more quickly within the monocytes,” says the researcher. In response to the increasing viral load, monocytes release large amounts of proteins (cytokines) that aggravate the body’s inflammatory response, and consequently the disease.

Kika Carvalho
The virus can infect cells in the pancreas and the liver, which control blood glucose (sugar) levels, in some cases causing diseases such as diabetesKika CarvalhoThis phenomenon may help to explain why COVID-19 is more severe in individuals with diabetes, who have high blood glucose levels. In recent months, however, researchers have observed an increasing number of cases where people with no history of diabetes have experienced an increase in glucose levels after becoming infected with SARS-CoV-2. One explanation suggested by Mori is that the virus infects cells in the pancreas, liver, and adipose tissue, which control glucose levels. It is too early to know for sure whether there is a causal relationship between these phenomena, but experts are not ruling out the possibility that in some cases, COVID-19 could lead to the onset of diabetes.
Another aspect that has doctors and researchers concerned is the complications caused by excessive blood clots (thrombi). Depending on where they form, thrombi can cause serious problems. They usually occur in veins deep inside the legs or pelvis, from where they can travel to the lungs. Clots originating in veins in the neck and chest can travel to the brain. If they are not broken down by medication, they can kill organs by restricting the supply of oxygen, which can be fatal to the patient. In more than 60 autopsies performed at FM-USP, Dolhnikoff and his team identified multiple thrombi in the small pulmonary arteries of most patients who died of COVID-19. The same was observed by Lacerda’s team in brain and heart autopsies of victims of the disease in Manaus.
It is believed that this is a consequence of SARS-CoV-2 infecting the cells that line the inner wall (endothelium) of the blood vessels. In many patients, this cellular invasion causes a series of changes in the blood clotting mechanism, leading to hypercoagulability, thus increasing the likelihood of thrombi that can cause heart attacks and hemorrhages in the heart and brain (see article). “The consequences of these complications are the same as any vascular accident,” says Oliveira, from the Emílio Ribas Institute.
The symptoms caused by the novel coronavirus infection and the subsequent complications are also associated with another important phenomenon. In many cases, the virus causes a strong and systemic inflammatory response, triggered by cytokines. “This overreaction by the immune system causes the immune cells to attack some of the healthy tissue around the affected area, increasing the damage to the affected organ and compromising its function,” explains Alves-Filho. It is a battle between the patient and their immune system. Sometimes, it occurs in vital organs such as the kidneys, affecting their ability to filter blood. “The incidence of acute kidney damage in patients hospitalized with COVID-19 is 36%,” highlights biochemist Adriana Girardi, from the teaching hospital (Hospital das Clínicas) at FM-USP. According to her, for more severe cases of the disease involving patients in ICU, this figure is as high as 90%. “Two of my patients now need hemodialysis, and they probably will for the rest of their lives,” comments Dalcolmo.
It is not yet known why some people’s immune systems react so strongly while others remain more focused on the infected cells. It may be linked to each individual’s genetic makeup or the presence of pre-existing conditions, which constantly expose the immune system to inflammatory molecules. This could compromise the body’s defense against other diseases, such as COVID-19. “Patients admitted to the ICU are at risk of secondary bacterial infections, which tend to exacerbate the body’s inflammatory response even further,” says Alves-Filho. “We are getting to know the disease as it spreads. It is like changing the tire of a moving car,” summarizes Dolhnikoff.
Republish