Knowing the number of people infected with a disease is vital in estimating the size of a pandemic and responding with appropriate public-health policies. In the case of SARS-CoV-2, the virus that causes coronavirus disease 2019 (COVID-19), case numbers will determine whether lockdowns need to be imposed on cities or entire regions, how long social-distancing measures should continue, and when these measures—which are as necessary as they are unpopular—can be relaxed. Case counts should ideally be derived from large-scale testing. When this is not possible, clinical diagnoses of the disease can provide an important, even if less precise, measure of the infected. Case data is fed into reporting systems, of which in Brazil there are two: the e-SUS Epidemiological Surveillance System (e-SUS VE) and the Epidemiological Influenza Surveillance Information System (SIVEP-GRIPE). The first is used to report mild cases of the disease that do not require hospitalization, while the second is for patients who have been hospitalized with Severe Acute Respiratory Syndrome (SARS), a condition caused by different viruses, including SARS-CoV-2. The SIVEP-GRIPE system allows users to track a patient’s history in the hospital, from admission to discharge.
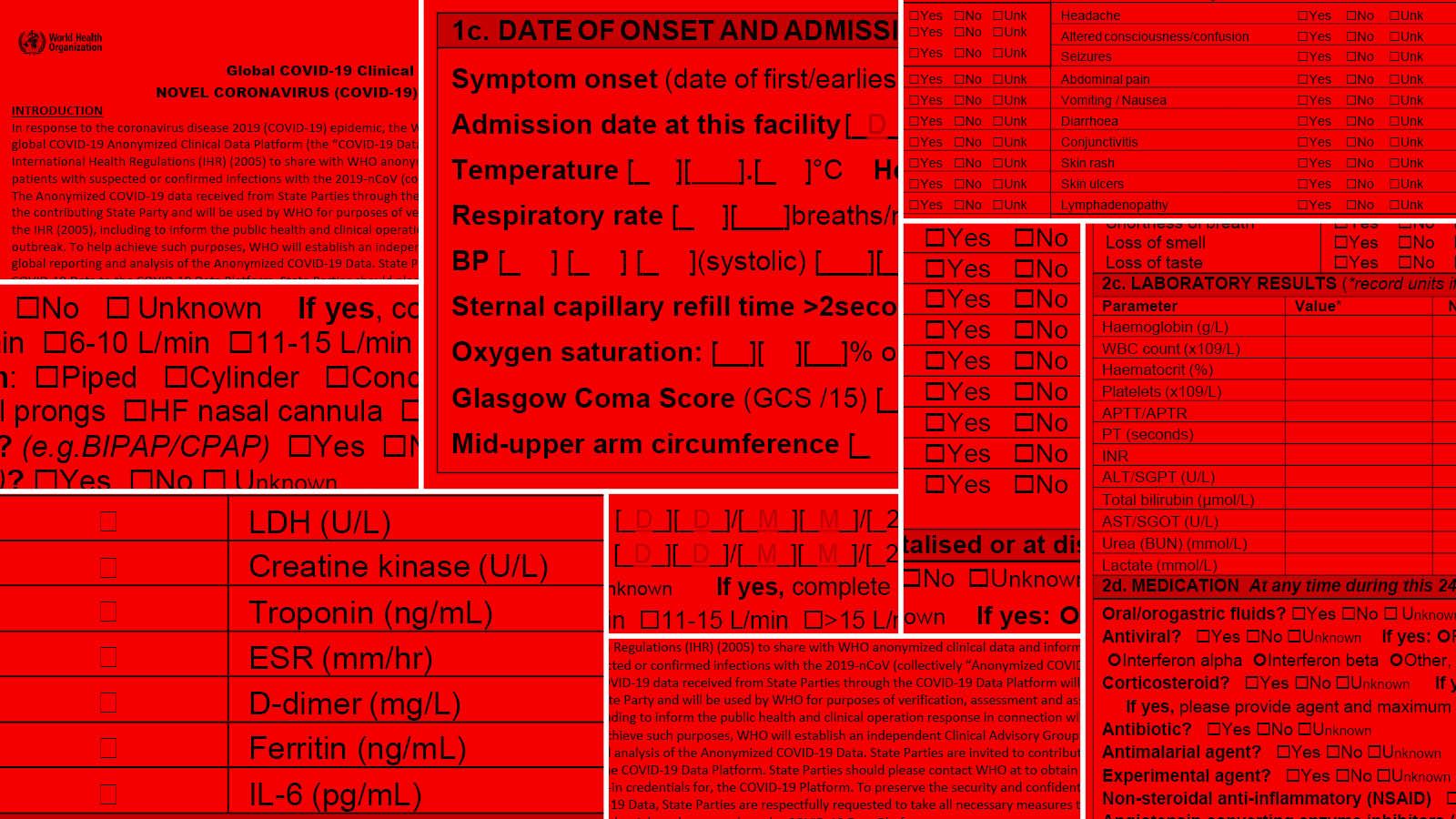
Case-reporting systems use case definitions—a set of standard signs and symptoms—to ensure that healthcare providers use a consistent protocol to identify patients. During pandemics, case definitions are recommended by the World Health Organization (WHO). For COVID-19, WHO developed a standard form for collecting patient data at three different points in the patient journey: on admission to the facility; on admission to the ICU, if applicable; and on discharge. Under WHO guidelines, cases are reported as either confirmed (by laboratory testing), suspected (when clinical symptoms are present, but patients have not been tested), or probable (when individuals with symptoms have been in contact with confirmed patients, have traveled to locations with community transmission, or have been inconclusively tested).
These guidelines have been used as a basis for reporting systems around the world, including in Brazil, but with each country defining its own disease surveillance criteria. Here, only confirmed cases are included in infection and death counts, whereas other countries also include probable cases.
COVID-19 is a reportable disease in Brazil, along with 50 other conditions—such as syphilis, meningitis, and measles—that require surveillance by health authorities in order to prevent or contain outbreaks. New cases of these diseases are normally required to be reported in the Reportable Disease Information System (SINAN). The tool, launched by the Ministry of Health in the early 1990s, has a database that has helped to inform policies for preventing and containing outbreaks of different diseases. But because it relies on uploads from municipal and state health departments, the SINAN system is not particularly useful in situations requiring data to be updated in real time.
As a result, COVID-19 case reporting has been migrated from SINAN to dedicated systems that can be updated online without requiring file uploads. One is e-SUS VE, a database launched on March 23 to report suspected, confirmed or ruled-out cases of “influenza-like illness,” a collection of symptoms that include feverishness or fever accompanied by a cough, sore throat, runny nose, or difficulty breathing. “One of the problems with this platform is that after suspected cases are catalogued, hospitals are unable to update epidemiological records with testing results. Only departments of health and a handful of other institutions are able to do this,” says epidemiologist Ana Freitas Ribeiro, who heads the Institute for Infectious Diseases’ Epidemiology Service at Emílio Ribas Hospital, and is a professor of graduate studies at Universidade Nove de Julho (UNINOVE). The Ministry of Health’s instructions that people with mild symptoms stay at home and monitor the progression of their symptoms has also affected e-SUS VE reporting. “Although on the one hand this strategy was necessary to prevent the healthcare system from being overwhelmed and focus efforts on treating serious patients, it also resulted in significant underreporting,” says Ribeiro.
The e-SUS VE platform is used to report patients with mild flu symptoms only; confirmed or suspected cases of COVID-19 that have evolved to SARS are reported in the SIVEP-GRIPE database. Hospitals across the country have been required to report patients hospitalized with SARS since the influenza A (H1N1) pandemic in 2009, and now again with the SARS-CoV-2 pandemic. The platform can be used to track a patient’s entire journey through the hospital from admission to outcome. José Olímpio Moura de Albuquerque, a public-health physician at the Health Surveillance Office at the Municipal Department of Health (COVISA-SMS) of São Paulo, explains that the system has been used to report COVID-19 cases that have progressed to SARS since community transmission was confirmed on March 20. “This means that as long as hospitals have beds available for admission, most severe COVID-19 cases—accounting for 10% to 20% of total cases of the disease—will be reported in the system,” he says.
But if patients stay at home due to overwhelmed hospitals, severe cases will no longer be reported in the SIVEP-GRIPE database. With COVID-19 beds reaching full capacity in some regions, the progression of the pandemic will need to be tracked indirectly using data on waiting lines for public hospital beds. Both e-SUS VE and SIVEP-GRIPE provide patient profile information including sex, race, age, and address, making it possible to detect patterns of disease spread, explains Albuquerque. “In São Paulo, for example, coronavirus spread showed a radiating pattern outward toward the suburbs,” he says.
Compulsory reporting deadlines vary depending on how contagious the disease is. For COVID-19, suspected cases must be reported within 24 hours. Epidemiologist Carla Gianna Luppi, a professor at the Federal University of São Paulo (UNIFESP) who heads the São Paulo State Referral and Training Center for Sexually Transmitted Diseases and AIDS (DST/AIDS), explains that to meet the deadline, hospitals are entering patient data in the SIVEP-GRIPE system immediately on admission, while collecting samples of biological material to identify the virus causing the respiratory disease. When they get the test results back from the lab, they then update the patient’s health record in the SIVEP-GRIPE system.
Despite hospitals’ efforts to report suspected cases as promptly as possible, many have faced operational constraints. Ribeiro, of Emílio Ribas Hospital, notes that some healthcare providers still use paper forms that are sent in mail pouches to regional health surveillance offices to be entered on the SIVEP-GRIPE platform. In São Paulo, 27 hospitals still have paper-based reporting systems.
After suspected cases are reported on admission, hospitals still need to identify the underlying respiratory virus before committing the report in the system, either confirming infection by SARS-CoV-2 or another agent, or classifying the case as nonspecified, when the cause of the disease cannot be determined. Until this happens, the case is kept open and is not counted in the official statistics of state health departments and the Brazilian Ministry of Health. All suspected cases, however, are included in epidemiological bulletins published by the Ministry of Health.
It can take up to 60 days from patient admission to confirm infection by COVID-19—this is the maximum period allowed under a 2005 directive from the Ministry of Health for hospitals to close cases in the system. Testing results are the primary—but not the only—aspect of a patient’s clinical status. The final diagnosis also includes information about the patient’s recovery or death. “COVID-19 hospitalizations can extend for two to three weeks, and we can only close the case in the system and confirm or rule out the disease after the clinical outcome—either discharge or death—is known,” says Ribeiro.
Investigating cases until closure can help to identify positive patients who initially tested negative for COVID-19 at an early stage, when the virus is difficult to detect, she explains. At Emílio Ribas, when a test comes back negative, the attending physician takes into account the time when the sample was collected, the results of other laboratory tests, the patient’s progression, and X-ray findings before entering a diagnosis in the SIVEP-GRIPE system. The Ministry of Health’s official count includes only those cases that have been confirmed by laboratory testing and reported through COVID-19 case-reporting systems, i.e. e-SUS VE and SIVEP-GRIPE.
Underreporting will be low for serious cases if the hospitals admitting COVID-19 patients have beds available for hospitalization, but there is a lag between the time a case is confirmed and when it is reported. Albuquerque, of COVISA-SMS, notes that one of the biggest constraints on decision-making is the case-reporting lag. “From the time a patient falls sick, seeks medical attention, and is admitted to the time the case is reported, at least 10 days will typically have elapsed depending on the hospital,” he estimates.

Alexandre Schneider / Getty Images
The Vila Formosa Cemetery in São Paulo, the largest in Latin America, saw a 50% increase in burials in May compared to the same month in 2019. Excess mortality provides a measure of the real impacts from the pandemic on healthcare systems
Alexandre Schneider / Getty Images“In general, a hospital will not have dedicated staff to enter patient data in disease-reporting systems. Even in previous years when the healthcare system was not overwhelmed as it is now, SARS cases could take as much as a month to be reported,” says Marcelo Gomes, a public-health researcher in the Scientific Computing Program at Oswaldo Cruz Foundation (FIOCRUZ) who manages the Infogripe SARS reporting platform in Brazil. Another problem, Gomes notes, is duplicate reporting of cases—a case can be reported in the e-SUS VE database when a patient receives care at an outpatient clinic, and then be reported redundantly in the SIVEP-GRIPE system if that same patient is later hospitalized. “Each system has its own database and they are not integrated together. Municipal and state authorities have worked to eliminate duplicate cases by going through patient records in the two systems to manually delete repeated records. But this requires time and staffing,” says Gomes.
The lack of integration between the SIVEP-GRIPE platform and state-run Central Public-Health Laboratories (LACEN) is an additional obstacle to effective COVID-19 reporting. These labs receive biological material from patients admitted to public hospitals and perform tests and repeat tests for detection and confirmation of SARS-associated coronavirus. Under optimal conditions, when there are no other samples in the queue to be processed, it takes three to five days to receive the results from an RT-PCR test, which can detect the virus directly in respiratory secretions. The results are then entered in the LACEN network Laboratory Management System (GAL). This system is not integrated with the SIVEP-GRIPE database. “Which means health professionals need to access the GAL system and manually enter the results in the patient’s record in the SIVEP-GRIPE system,” says Gomes. The Ministry of Health will only consider a case to be confirmed after the test results have been entered and the patient’s record has been committed on the platform.
In Brazil, total case counts have been estimated by university initiatives such as the COVID-19 Prevalence Survey (Epicovid19-BR) at the Federal University of Pelotas (UFPel), in Rio Grande do Sul, southern Brazil. The study estimated that approximately 2.5 million people had been infected as of May.
The Mortality Information System, available on the Federal Government’s DATASUS platform, contains data on all deaths occurring in Brazilian territory. Data is uploaded to the platform every two years, which means current data is for 2018—long before the first coronavirus cases were reported.
In addition to the SIVEP-GRIPE database, Brazil’s vital statistics website provides another source of information on the number of deaths caused by COVID-19 and by respiratory complications in general. One of the problems with this platform, says Benilton de Sá Carvalho, a professor in the Department of Statistics at the University of Campinas (UNICAMP), is that the timing of data entry varies widely from one vital records office or municipality to another. While some enter the data on the platform immediately on receiving a death certificate, others can take up to three years. “This does not mean the data is inaccurate, only that it is incomplete,” says Carvalho. Statistical analysis, he says, can provide a means to estimate the total quantity of a given phenomenon, even with incomplete data. However, this requires information on what portion of the content is lacking—and this information is unavailable. Another limitation, says Carvalho, is that the platform lacks tools to retrieve snapshots of data from specific periods in the past.
Brazilian newspaper Folha de S.Paulo reported on May 14 that around half a million deaths had vanished from the records on the portal. According to the Brazilian Association of Vital Statistics Offices (ARPEN), the deleted records were duplicates in the Rio de Janeiro database. “Duplicate data is a common problem in large data sets. But death records are supposed to be individually unique, as they are confirmed against the deceased person’s identity number, social security number, and employment records,” says Carvalho, noting, however, that when these documents are unavailable, as is often the case, vital records offices are unable to crosscheck data, leading to duplicate records. The existence of duplicate data means the figures on the portal cannot be relied on as a lower-limit death count. “We assumed that with each update the number of deaths would necessarily rise, but we now know they could also decrease if further duplicate records are purged,” he explains.
According to Epidemiologist Paulo Lotufo of the University of São Paulo School of Medicine (FM-USP), data from the funerary office in São Paulo may provide the most accurate picture of how lethal COVID-19 is. “The funerary office delivers a copy off all death certificates it receives to the Municipal Health Department’s Mortality Information Improvement Program [PRO-AIM] within 24 hours. This means information on deaths in the municipality is updated virtually in real time,” says Lotufo. In analyzing the information in the database, Lotufo found that the number of deaths from natural causes between January and February this year was similar to the average for the previous five years. But in March there were 743 more deaths than the average for that month between 2015 and 2019; 277 of those 743 deaths were officially reported as COVID-19 deaths. This is known as excess mortality and provides a measure of the real impacts from the pandemic on the healthcare system.
Using the same methodology, the European Mortality Monitoring (EuroMomo) project analyzed the mortality curve from March 23 to April 19 in 24 European countries, and found that 100,000 more people died than on average in the previous five years.
Case reporting in the UKIn England, National Health Service (NHS) laboratories use an electronic system to report positive SARS-CoV-2 cases directly to Public Health England, an agency of the Department of Health and Social Care.
Gwenda Hughes, who heads the STI Surveillance Section at the National Infection Service, says other countries in the United Kingdom (UK) use similar systems. Hughes, in an email interview with Pesquisa FAPESP, explained that early in the pandemic, only deaths from COVID-19 that occurred at hospitals were counted.
Since the end of April, however, all deaths of individuals who test positive for the disease have been counted in the official statistics, including not only those who have lost their lives in a hospital setting, but also patients who have died at home and in nursing homes. “Data from the Office for National Statistics includes both confirmed and suspected cases of COVID-19 based on death certificates. Deaths are reported with an 11-day lag because they first need to be certified, recorded, and processed,” she says.
Republish